seemabmaida
Maharishi Markandeshwar University - Mullana Campus
1 Follower
0 Following
0 Helped
seemabmaidaLv2
6 Mar 2023
Answer: Bio means life and logy means study. Study of life .There are differen...
seemabmaidaLv2
19 Jan 2022
Answer: The substance HOBR is consideredA) a weak Arrhenius acid.B) a weak Arr...
seemabmaidaLv2
19 Jan 2022
Answer: Arrhenius acid formulaNameHNO3Nitric acidHClO4Perchloric acidH3PO4Phos...
seemabmaidaLv2
17 Jan 2022
Answer: 1.Step-by-So we are asked to add the falling two meter seas together. ...
seemabmaidaLv2
17 Jan 2022
Answer: 1.Your child (or grandchild) might be just a two-year-old, but it's no...
seemabmaidaLv2
17 Jan 2022
1.Answer: In the description section underneath the overview per the particula...
seemabmaidaLv2
17 Jan 2022
Answer: 1.PH3 is one of the most misunderstood chemical compounds due to its p...
seemabmaidaLv2
17 Jan 2022
Answer:You must be able to combine your knowledge of molecular shapes and bond...
seemabmaidaLv2
17 Jan 2022
Answer: 1.B 2.C
seemabmaidaLv2
17 Jan 2022
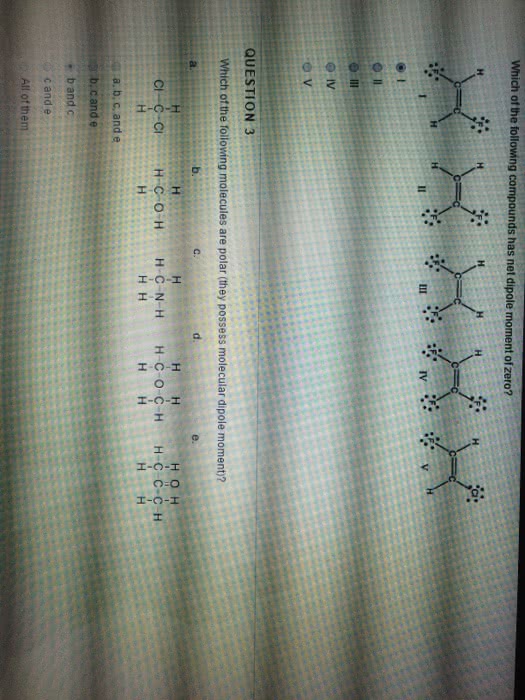
Answer: A CO2 molecule contains two polar bonds, but the net dipole moment is ...
seemabmaidaLv2
17 Jan 2022
Answer:
seemabmaidaLv2
17 Jan 2022
Answer: The octet rule refers to the tendency of atoms to prefer to have eight...
seemabmaidaLv2
16 Jan 2022
Answer:
seemabmaidaLv2
14 Jan 2022
Answer: 1.Vanadyl nitrate.it is an inorganic compound in the vanidium in the o...
seemabmaidaLv2
14 Jan 2022
Answer: List of Basic Chemistry Apparatus Safety goggles and safety equipment ...
seemabmaidaLv2
14 Jan 2022
Answer : A and B are the products obtained from thesodium borohydride reductio...