CHEM 134 Lecture Notes - Lecture 9: Svante Arrhenius, Activation Energy, Viscosity
33 views5 pages
5 Feb 2018
School
Department
Course
Professor
Document Summary
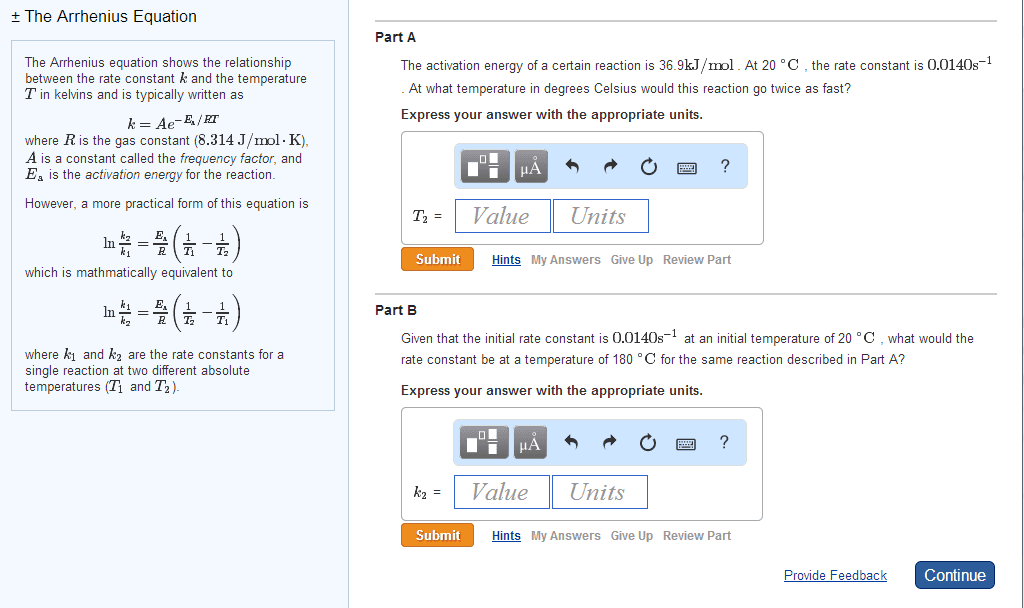
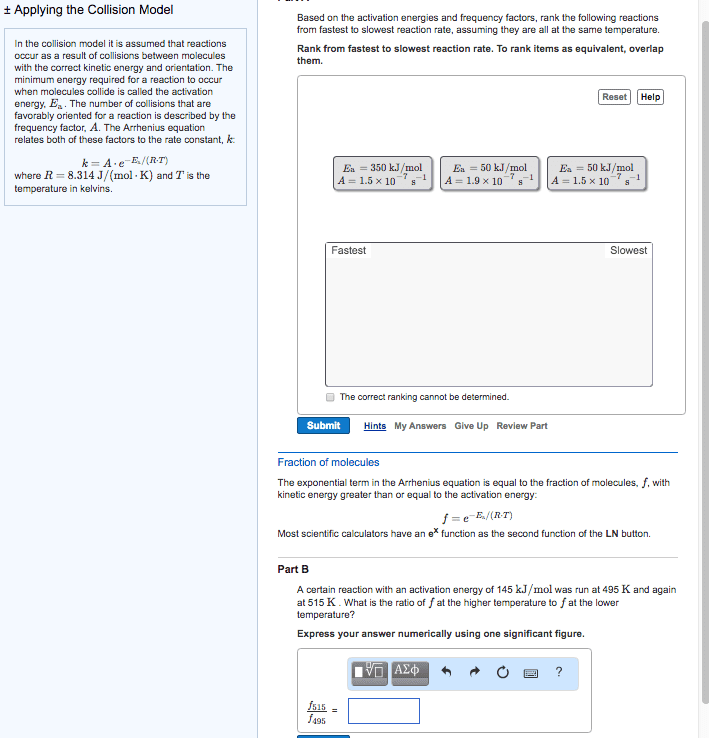
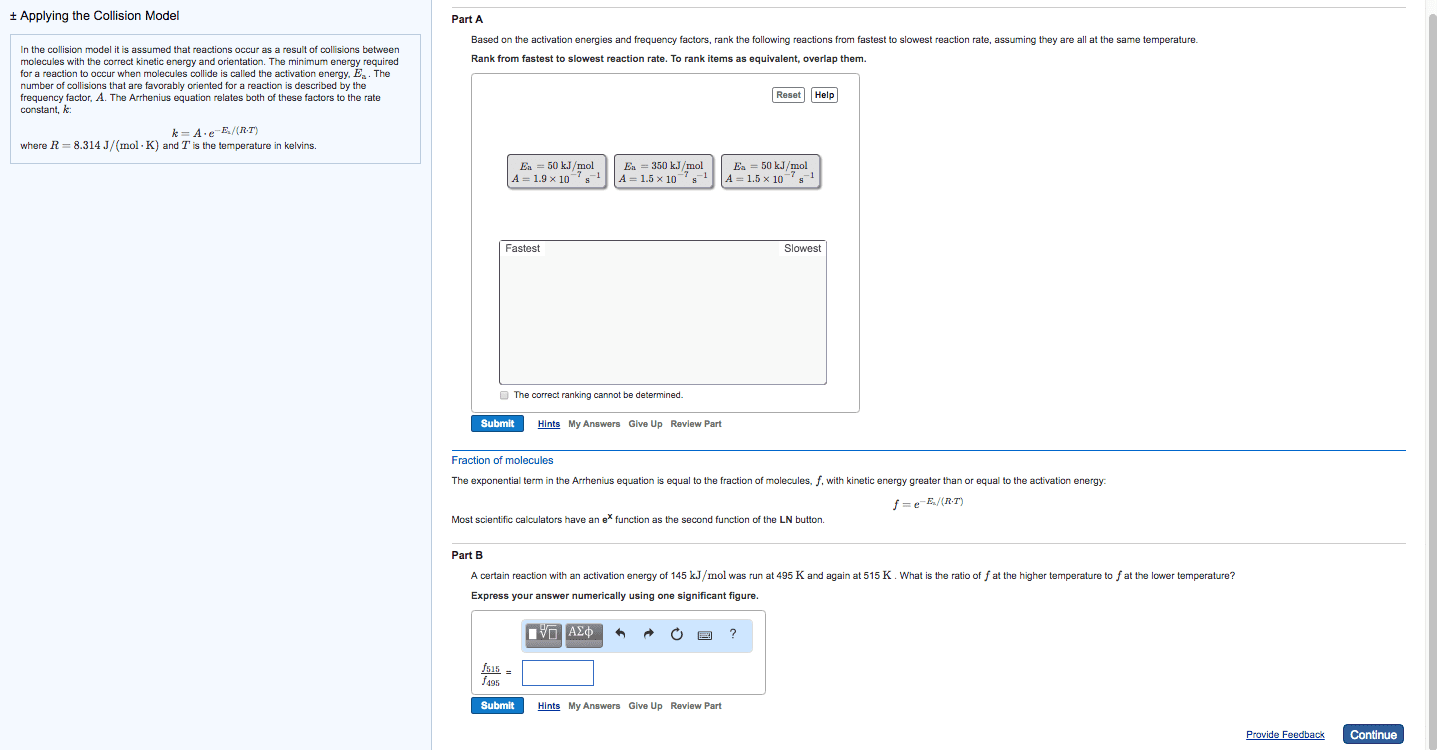
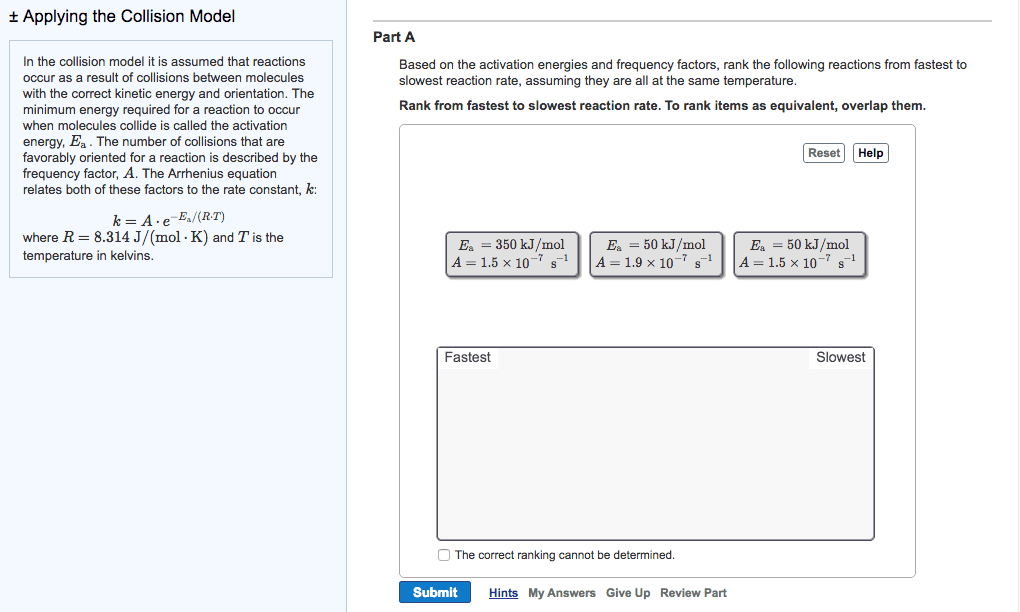
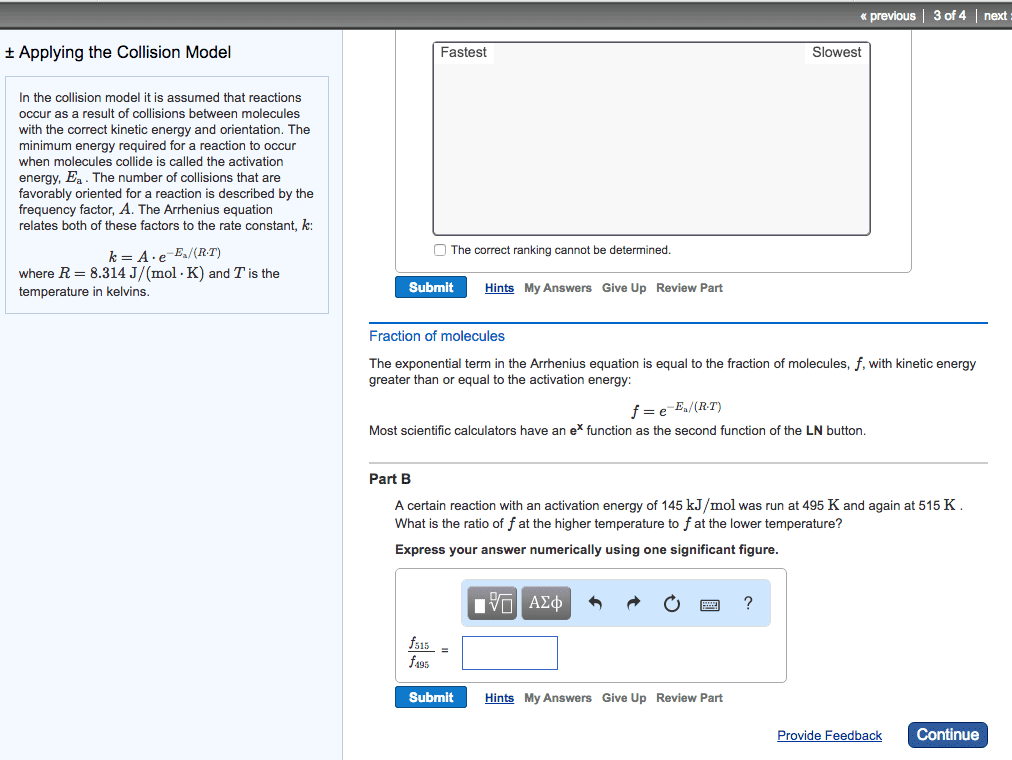
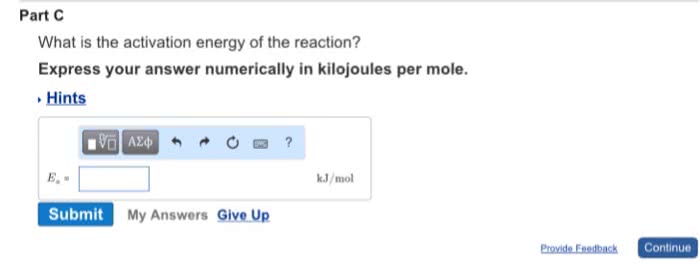
Increasing the temperature of a system increases the average kinetic energy of its constituent particles. As the average kinetic energy increases, the particles move faster, so they collide more frequently per unit time and possess greater energy when they collide, causing increases in the rate of the reaction. Rate of all reactions increases with increasing temperature and decreases with decreasing temperature. (2) the activation energy (ea ), and. If the arrhenius equation is written for two temperatures, t2 and t1 with t2 >t1 . Consider the rate of a reaction for which ea=50 kj/mol, at 20oc (293 k) and at. For reactions that have an ea 50 kj/mol, the rate approximately doubles for a 100c rise in temperature, near room temperature. The rate-law expression is known to be r=k[icl][h2 ]. The nature of the solvent can affect the reaction rates of solute particles. Solvent viscosity is also important in determining reaction rates.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232