CHEM102 Lecture Notes - Lecture 5: Arrhenius Equation, Collision Frequency, Collision Theory
117 views4 pages
24 Sep 2017
School
Department
Course
Professor

32
CHEM102 Full Course Notes
Verified Note
32 documents
Document Summary
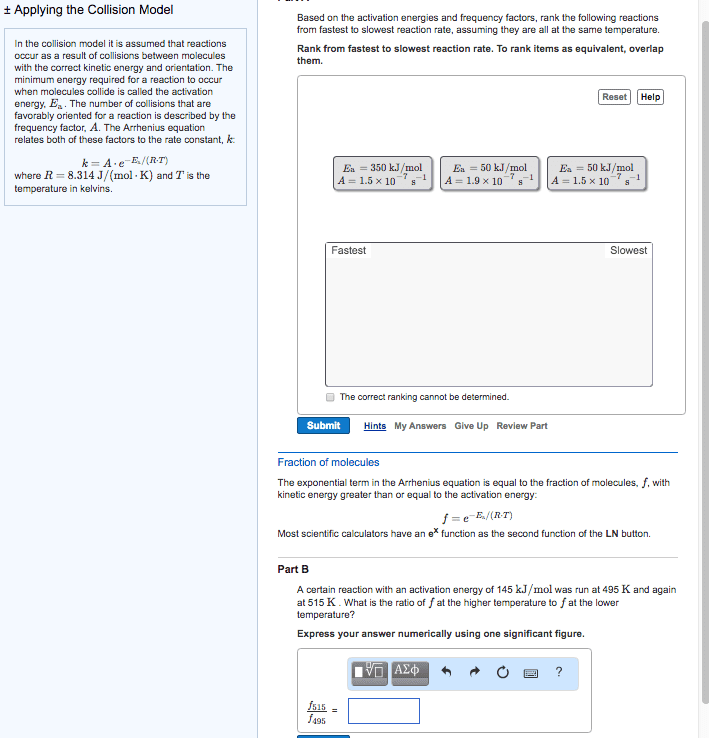
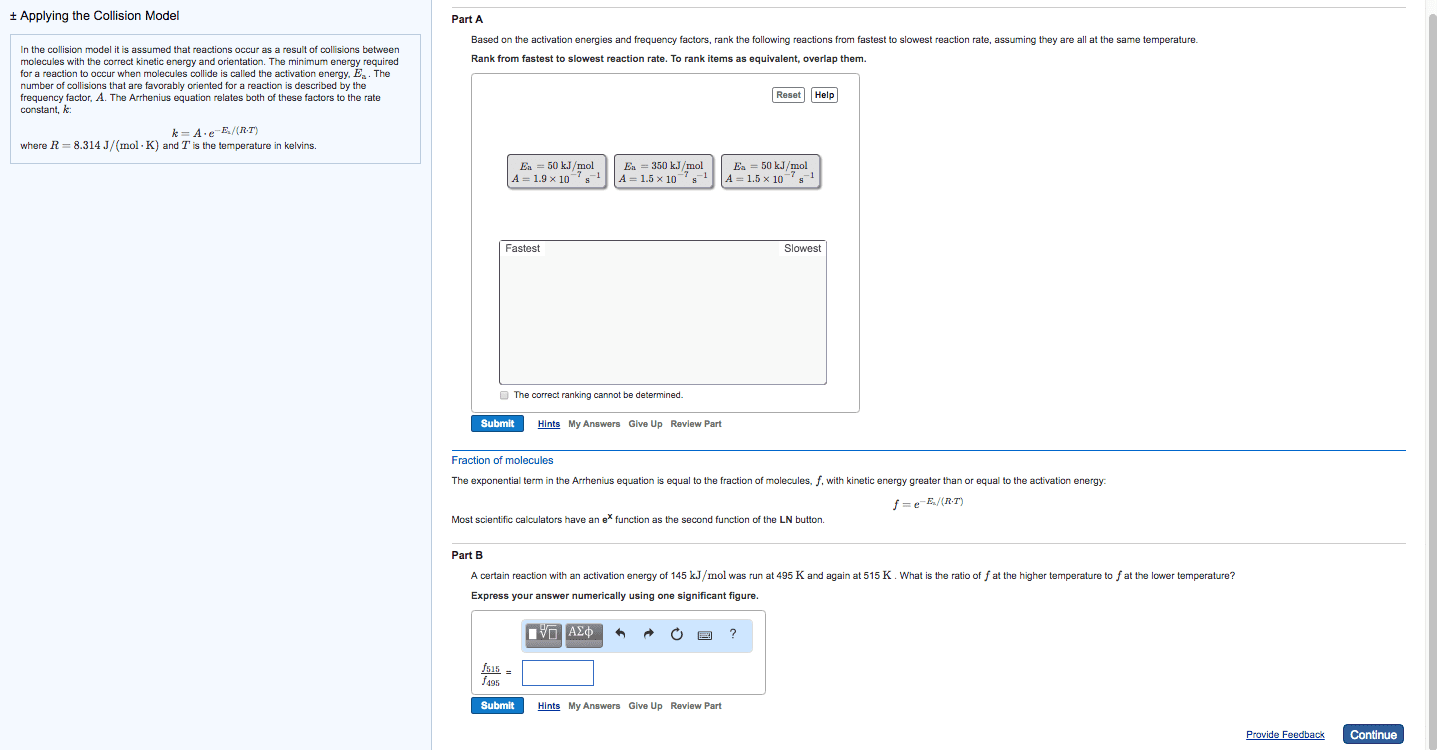
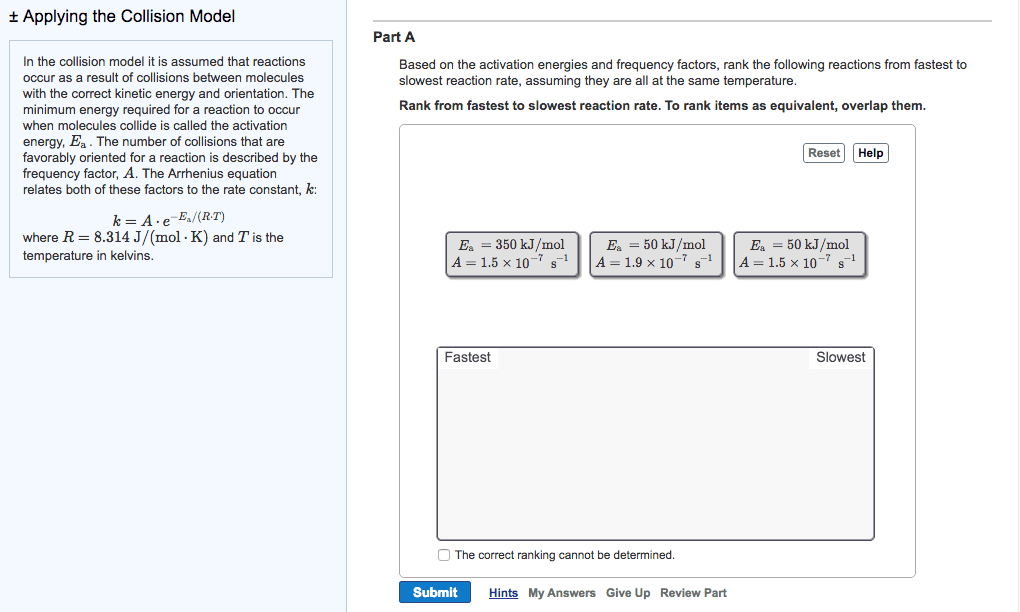
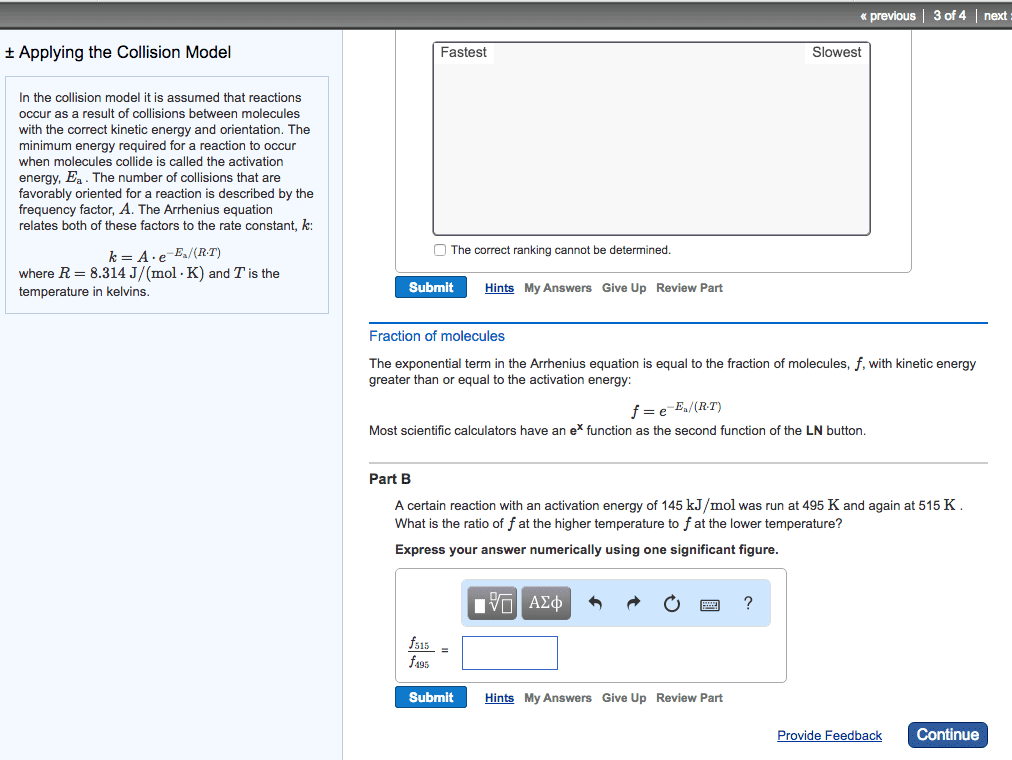
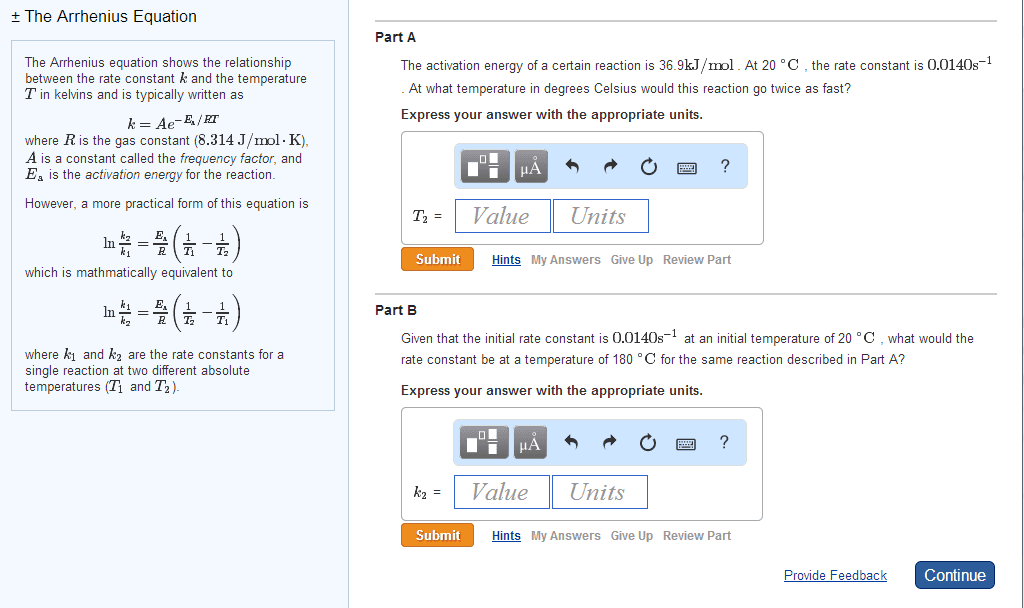
There is an energy barrier that the molecules must overcome. Rate of reaction is sensitive to change in temperature. In most reactions: the change in rate constant, k to the temperature (k) is exponential, for many reactions, an increase of 10oc in temperature doubles the reaction rate. Put forward by arrhenius in 1889, it is in the form of. Equation links the rate constant k with activation energy, ea and temperature, t k = ae-ea/rt. A = arrhenius constant and represents collision frequency factor. (how easy do the molecules collide with each other. ) Ea = activation energy of the reaction / j mol-1 or kj mol-1 e = base of natural logarithm and is equal to 2. 718. R = gas constant and is equal to 8. 314 j k-1 mol-1. The minimum energy the molecules must possess for a chemical reaction to take place is called the activation energy.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232