CHEM 001B Lecture Notes - Lecture 21: Elementary Reaction, Rate Equation, Reaction Step
69 views2 pages
Document Summary
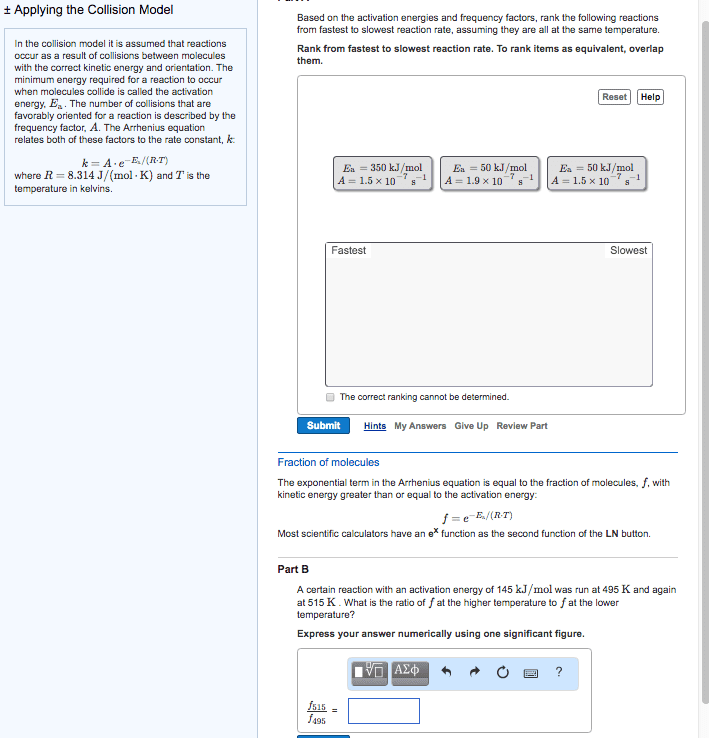
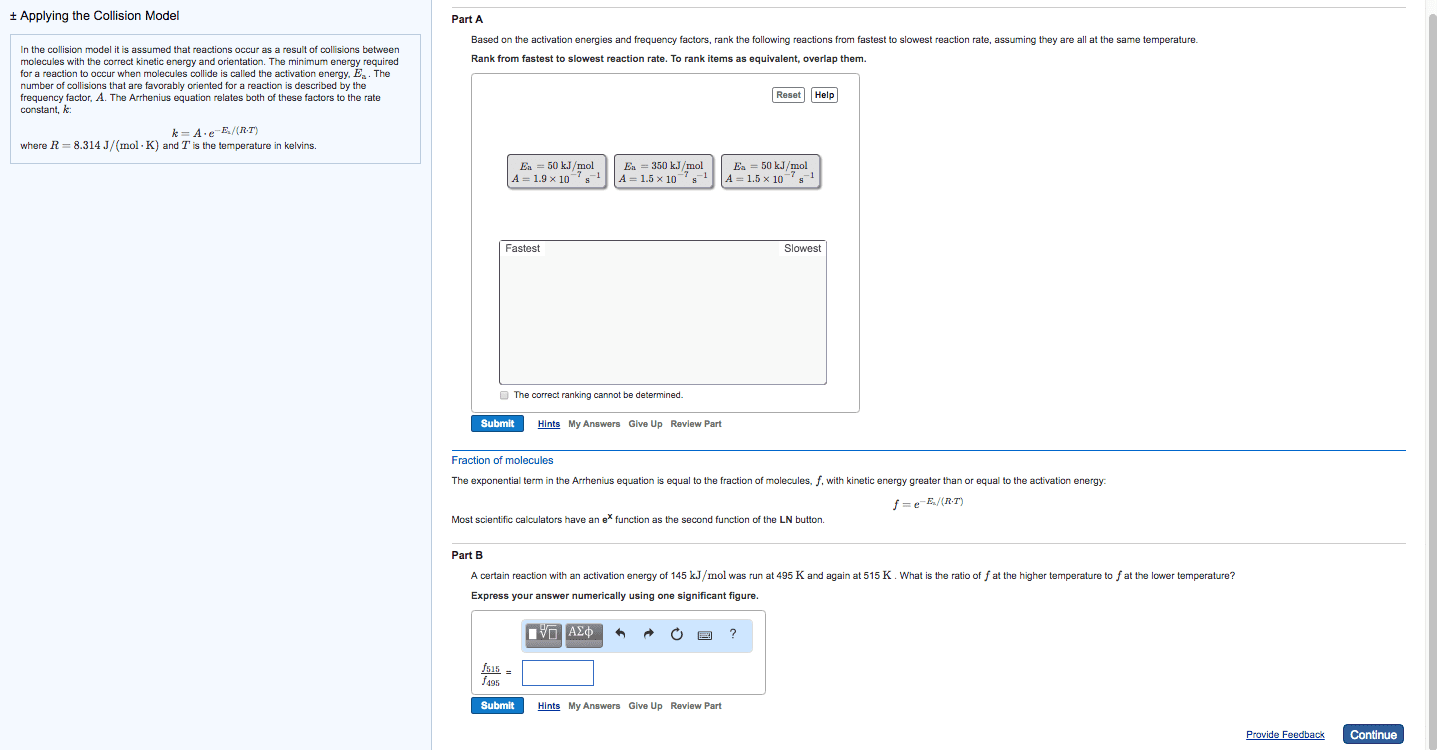
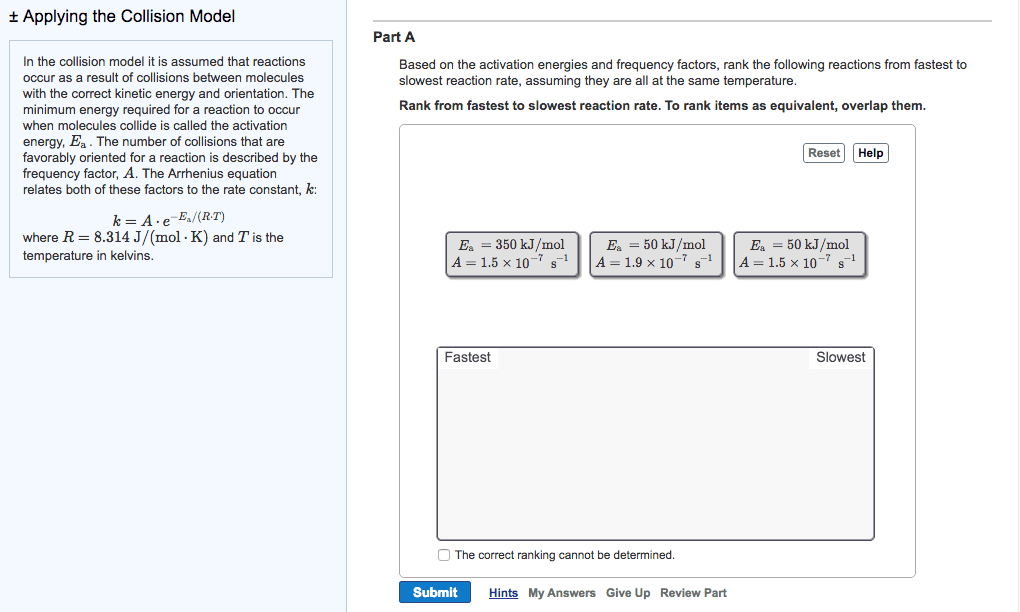
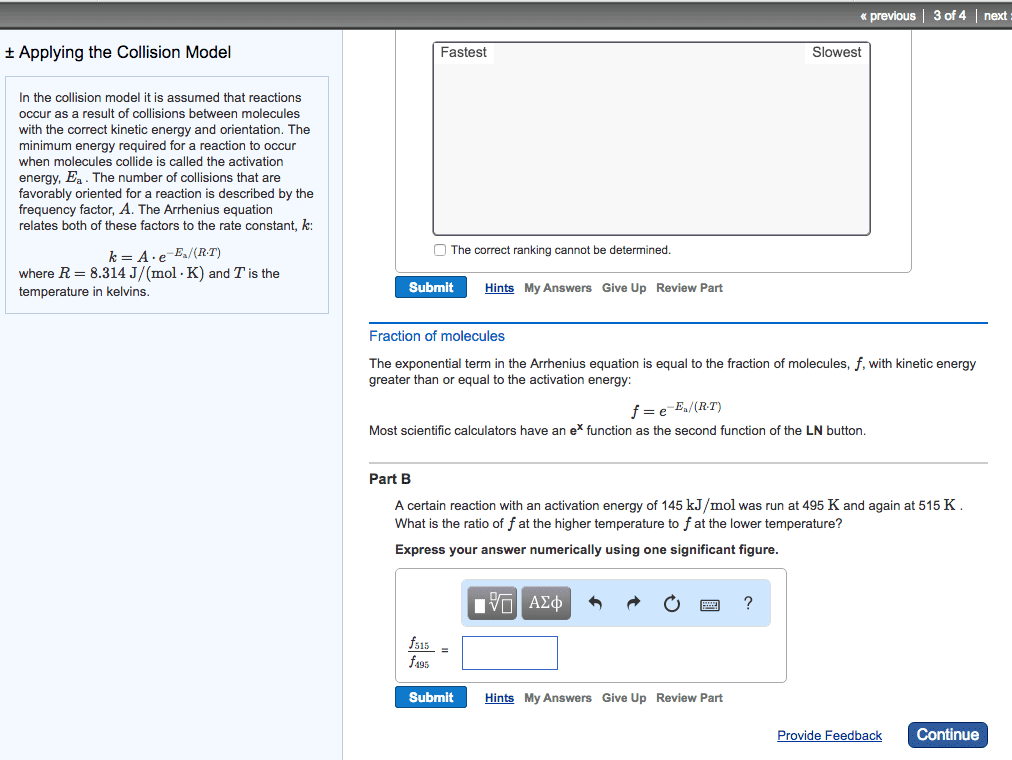
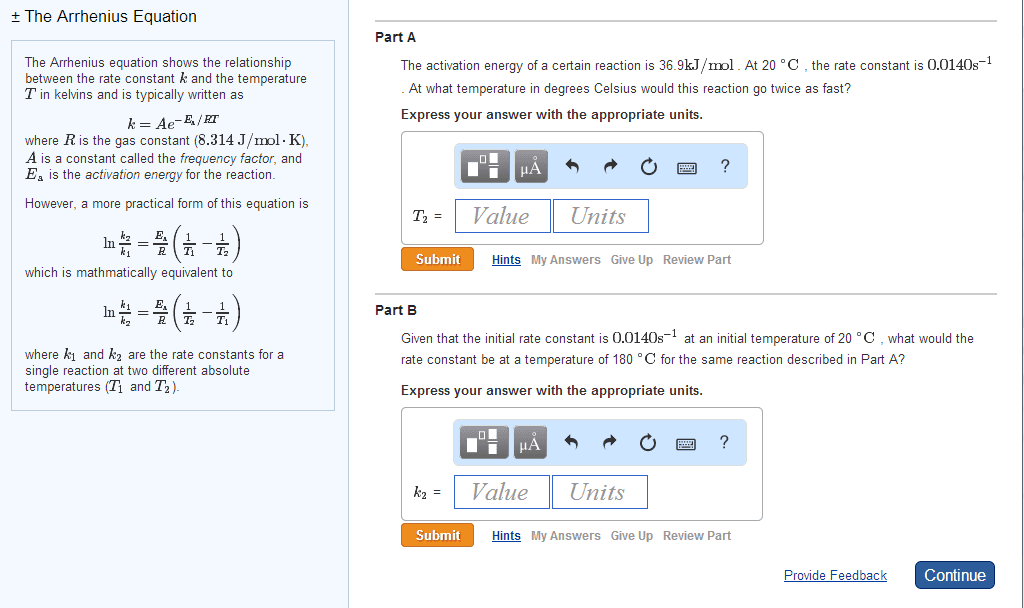
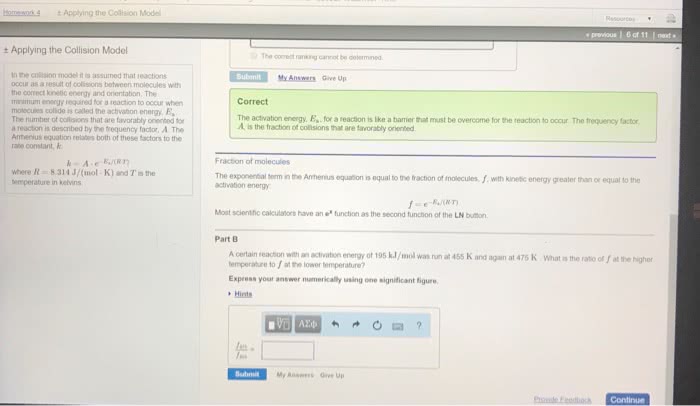
Increased temperature increases kinetic energy of molecules and. The minimum energy of molecular collisions required to break bonds in reactants, leading to formation of products. These factors are incorporated into rate constant k by arrhenius equation. R = universal gas constant [in j/(mol*k)] Includes frequency of collisions and an orientation factor. As temperature increases, the fraction of collisions exceeding this minimum energy also increases, leading to an increase in k and an increase in the rate of the reaction. Low e a = large k, fast forward reaction. High e a = small k, slow reverse reaction. Consider determination of k at two different t: The compound 1, 1-difluoroethane decomposes at elevated temperatures to give fluoroethylene and hydrogen fluoride: At 460 , k = 5. 8 x 10 -6 s -1 and e a = 265 kj/mol. Reaction mechanisms molecular level; must be consistent with the rate law for the reaction.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232