CHE 106 Lecture Notes - Lecture 35: Sigma Bond, Pi Bond, Vsepr Theory
CHE 106 verified notes
35/42View all
Document Summary
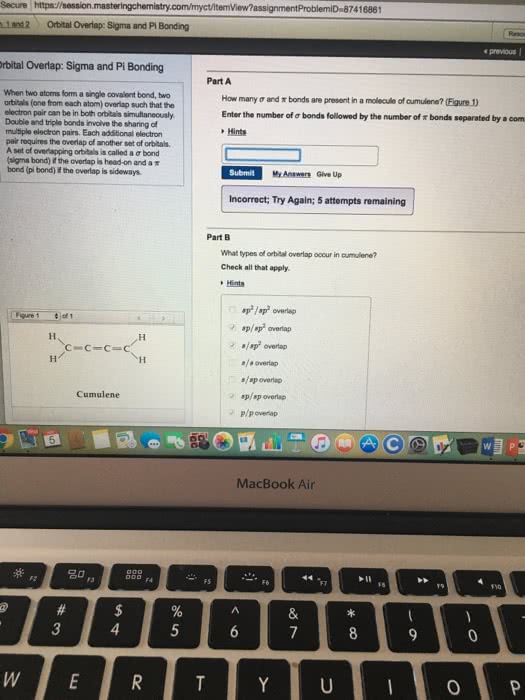
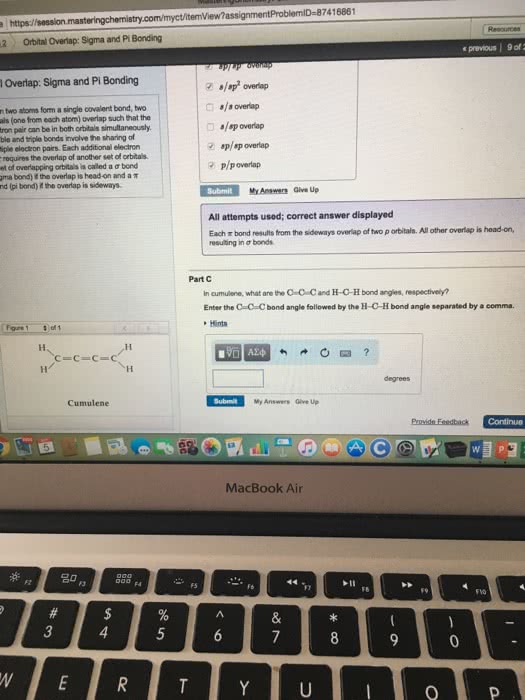
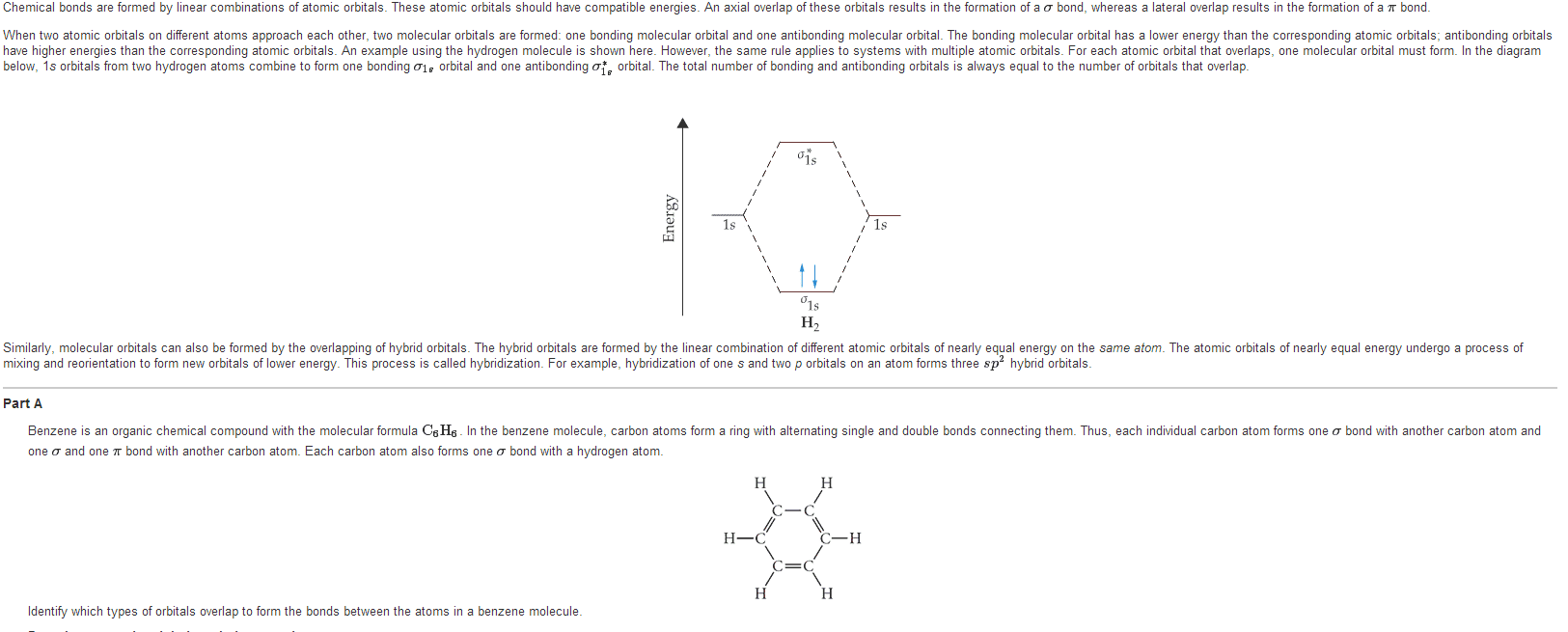
Vsepr: number of bonds formed by an atom=unpaired electrons, exception: carbon (2 unpaired electrons, form 4 bonds) Hybrid orbitals: form by mixing of atomic orbitals to create new orbitals of equal energy, called degenerate orbitals; process called hybridization: number of hybrid orbitals=sum of atomic orbitals. Hybridization: s3 geometric arrangement of hybrid orbitals same as vsepr theory sigma bond: head-to-head overlap of orbitals, cylindrical symmetry about internucleus axis pi bond: side-to-side overlap, electron density above and below internucleus axis, weaker bond.