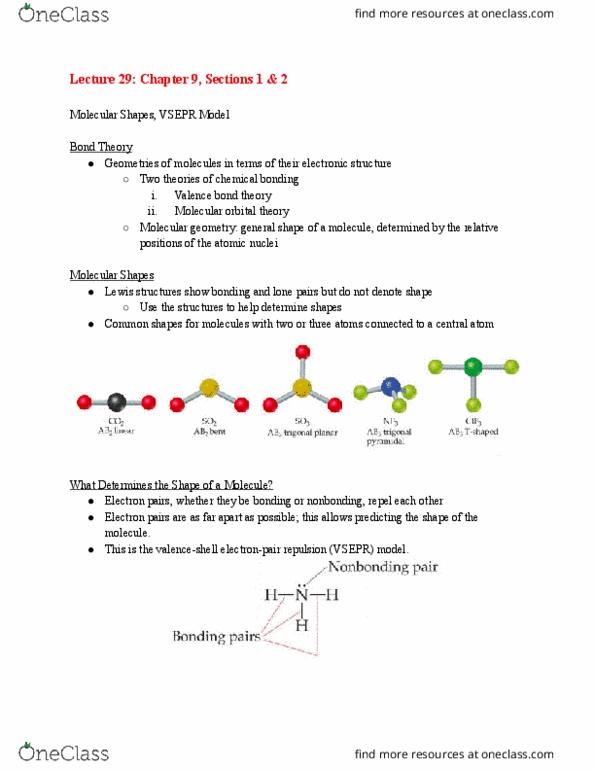
8 which of the following solids would have the highest melting point? ( 1) KI (2) KBr (3) KCI (4) KF 9. What type of chemical bond holds the atoms together within a water molecule? (1) polar ionic bond(3) non polar covalent bond (4) coordinate covalent bond 10. Which of the following covalent bonds is the most polar (i.e., highest percent ionic character)? (1) A -CI (2) Si- (3)Al- (4) SI-Ci 11. The total number of valence electrons in the ion SO is (1)32 (2)28 (3)30 (4) 26 12. The Lewis structure reveals a double bond in which of the following molecules? (1)N (2) Br (3) CS: (4) CaHe 13. The Lewis structure reveals an unpaired electron (free radical) in which of the following species? () NO (2)N:O (3) NO (4) NO 14. The number of lone electron pairs in the CIO ion is (1) 12 (2)16 (3)4 (4)0 15. The formal charge on the bromine atom in BrOs drawn with three single bonds is (1)*2 16. Which of the elements listed below would most likely form a polar covalent bond when bonded to sulfur? 17. Give the number of lone pairs around the central atom and the molecular geometry of SeF(1)0 lone 18. According to Molecular Orbital Theory, two separate 1s orbitals interact to form what molecular orbital(l)? 19. The Lewis dot symbol for the S-ion is (2)-1 (1) K (2) Mg (3)C (4)o pairs, tetrahedral (2) 1 lone pair, square pyramidal (3) 1 lone pair, tetrahedral (4) 1 lone pair, distorted tetrahedron (seesaw) (1)oonly (2) Ï only (3) Ï and Ï. (4) Ï and Ï. 2- 20. Which of the following molecules has polar bonds but is a nonpolar molecule? (1) CO (2) Cla 21. Use VSEPR theory to predict the geometry of the SFe molecule. (1) trigonal bipyramidal (2) bent 22. The geometry of the hybrid orbitals about a central atom with sp hybridization is (1) linear (2) bent 23. Which of the following molecules have the same geometries? (1) CO2 and BeHa (2) SFa and CHe 24. According to the VSEPR theory, which one of the following species is linear? (1)HaS (2) SO2 25. Which of the following substances will display an incomplete octet in its Lewis structure? (1) CO2 26. Predict the geometry and polarity of the CS2 molecule. (1) linear, polar (2) linear, nonpolar (3) SO (4) HBr (3) trigonal planar (4) octahedral (3) trigonal planar(4) tetrahedral (3) CO2 and NO2 (4) N2O and NO2 (3) BFs (4) BeF2 (2) NO (3) Cl2 (4) BrCI (3) bent, nonpolar (4) bent, polar
Show transcribed image text 8 which of the following solids would have the highest melting point? ( 1) KI (2) KBr (3) KCI (4) KF 9. What type of chemical bond holds the atoms together within a water molecule? (1) polar ionic bond(3) non polar covalent bond (4) coordinate covalent bond 10. Which of the following covalent bonds is the most polar (i.e., highest percent ionic character)? (1) A -CI (2) Si- (3)Al- (4) SI-Ci 11. The total number of valence electrons in the ion SO is (1)32 (2)28 (3)30 (4) 26 12. The Lewis structure reveals a double bond in which of the following molecules? (1)N (2) Br (3) CS: (4) CaHe 13. The Lewis structure reveals an unpaired electron (free radical) in which of the following species? () NO (2)N:O (3) NO (4) NO 14. The number of lone electron pairs in the CIO ion is (1) 12 (2)16 (3)4 (4)0 15. The formal charge on the bromine atom in BrOs drawn with three single bonds is (1)*2 16. Which of the elements listed below would most likely form a polar covalent bond when bonded to sulfur? 17. Give the number of lone pairs around the central atom and the molecular geometry of SeF(1)0 lone 18. According to Molecular Orbital Theory, two separate 1s orbitals interact to form what molecular orbital(l)? 19. The Lewis dot symbol for the S-ion is (2)-1 (1) K (2) Mg (3)C (4)o pairs, tetrahedral (2) 1 lone pair, square pyramidal (3) 1 lone pair, tetrahedral (4) 1 lone pair, distorted tetrahedron (seesaw) (1)oonly (2) Ï only (3) Ï and Ï. (4) Ï and Ï. 2- 20. Which of the following molecules has polar bonds but is a nonpolar molecule? (1) CO (2) Cla 21. Use VSEPR theory to predict the geometry of the SFe molecule. (1) trigonal bipyramidal (2) bent 22. The geometry of the hybrid orbitals about a central atom with sp hybridization is (1) linear (2) bent 23. Which of the following molecules have the same geometries? (1) CO2 and BeHa (2) SFa and CHe 24. According to the VSEPR theory, which one of the following species is linear? (1)HaS (2) SO2 25. Which of the following substances will display an incomplete octet in its Lewis structure? (1) CO2 26. Predict the geometry and polarity of the CS2 molecule. (1) linear, polar (2) linear, nonpolar (3) SO (4) HBr (3) trigonal planar (4) octahedral (3) trigonal planar(4) tetrahedral (3) CO2 and NO2 (4) N2O and NO2 (3) BFs (4) BeF2 (2) NO (3) Cl2 (4) BrCI (3) bent, nonpolar (4) bent, polar