CHE 106 Lecture Notes - Lecture 17: Bond-Dissociation Energy, Bromine, Joule
CHE 106 verified notes
17/42View all
Document Summary
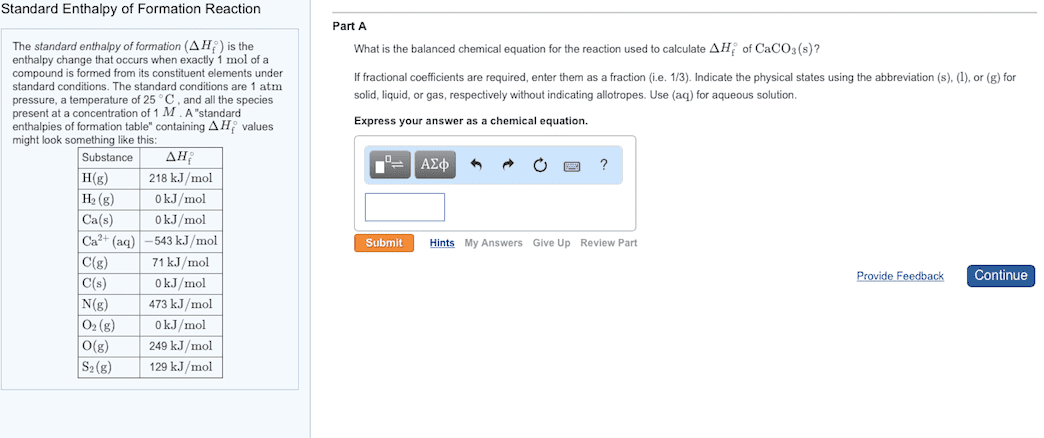
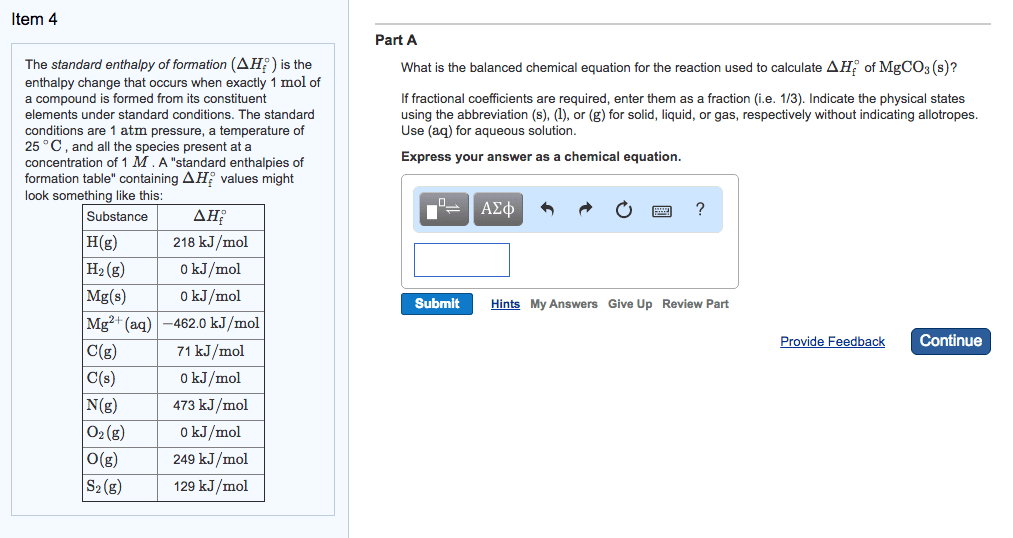
An enthalpy of formation, h f , is defined as the enthalpy change for the reaction in which a compound is made from its constituent elements in their elemental forms. The term standard state refers to the standard thermodynamic conditions chosen for substances: P = 1. 00 atm, t = 25. 0 c = 298. 2 k. The standard state for an element is the most stable form of the element for standard conditions. Oxygen o 2 (g), nitrogen n 2 (g), argon ar(g), bromine br 2 (l), iron fe(s), carbon. The enthalpy change for a reaction in which reactants are in their standard states is denoted h ( delta h zero or delta h naught ) The standard enthalpy of formation of a substance, denoted h f , is the enthalpy change for the formation of one mole of a substance in its standard state from its component elements.