CHE 106 Lecture Notes - Lecture 15: Enthalpy, Chemical Equation, Conversion Of Units
CHE 106 verified notes
15/42View all
Document Summary
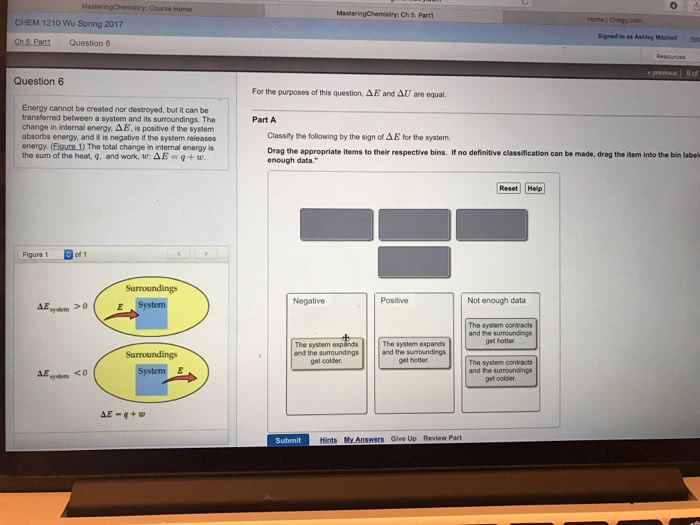
Chapter 5, sections 3 & 4, lecture 15. When energy is exchanged between the system and the surroundings, it is exchanged as either heat (q) or work (w). Q > 0, heat flows from surroundings into the system (endothermic) Q < 0, heat flows from the system to surroundings (exothermic) W > 0, work is done on the system (system is compressed) W < 0, work is done by the system (system expands) Change in e > 0, the system absorbed energy from the surroundings. Change in e < 0, the system released energy from the surroundings. Usually, a reaction takes place in an open vessel at constant pressure. It would be nice to have a state function whose change in value is equal to q for constant pressure processes. Enthalpy (h) is used to measure the flow of heat in chemical systems under a constant pressure (q p )