CHE 106 Lecture Notes - Lecture 15: Electric Potential Energy, International System Of Units, Thermodynamics
CHE 106 verified notes
15/42View all
Document Summary
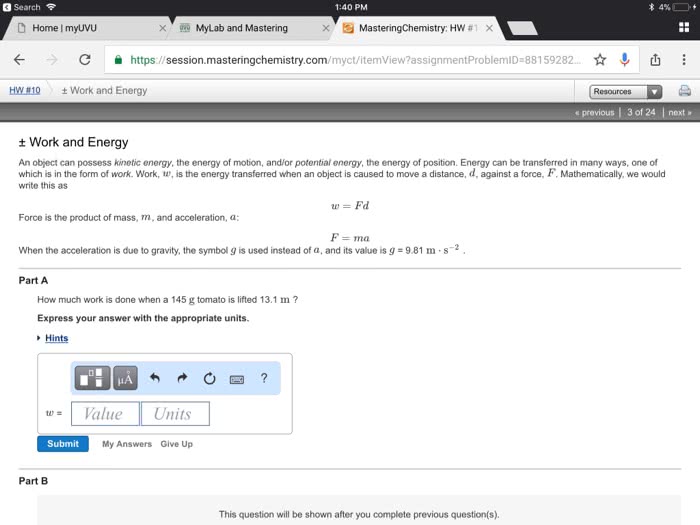
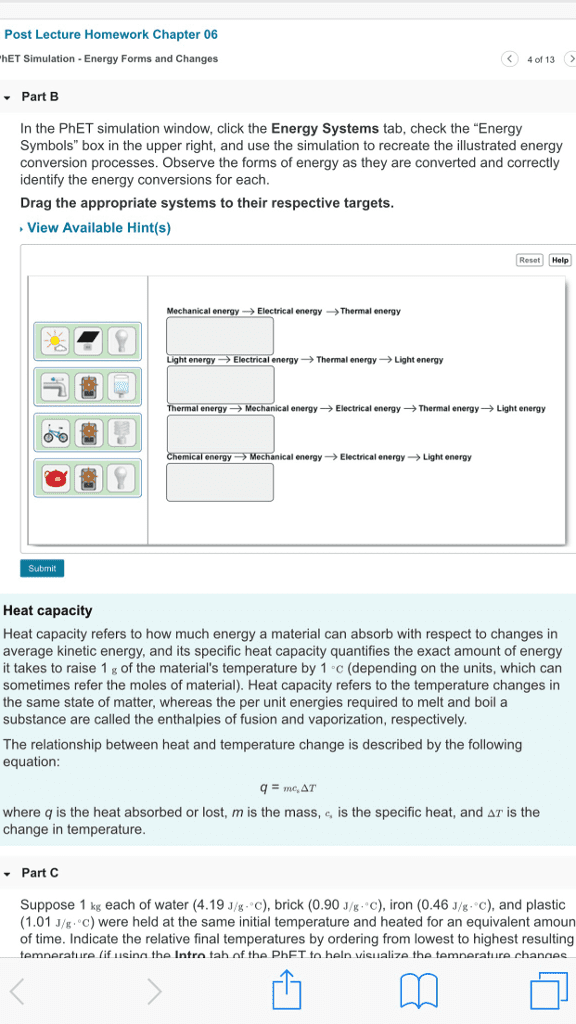
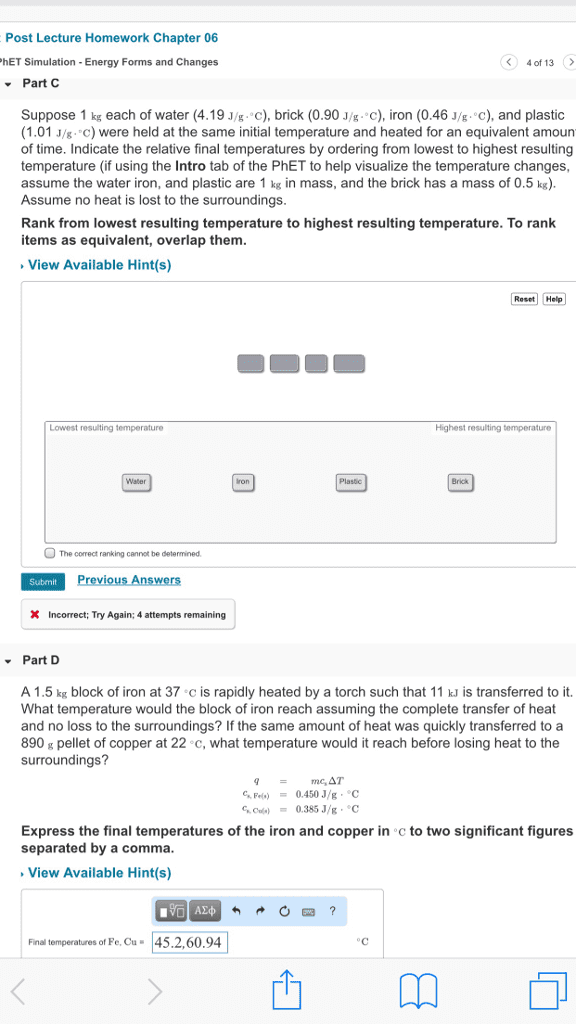
Thermodynamics: the science of the relationship between heat and other forms of energy. Thermochemistry: the study of the quantity of heat absorbed or evolved by chemical. In thermochemistry, heat energy can be found as a reactant or a product in chemical reactions reactions. Defined as the capacity to move matter: kinetic energy: energy associated with an object by virtue of its motion, an object of mass, m , velocity, v , has kinetic energy e k equal to i. i. E p = mgh: potential energy: the energy an object has by virtue of its position in a field of force, given mass, m , position, h , gravitational field, g. Internal energy: sum of kinetic and potential energies of the particles making up a substance. Most important form of potential energy in molecules is electrostatic potential energy, e el. Si unit of energy is joule (j) Electrostatic attraction is seen between oppositely charged ions.