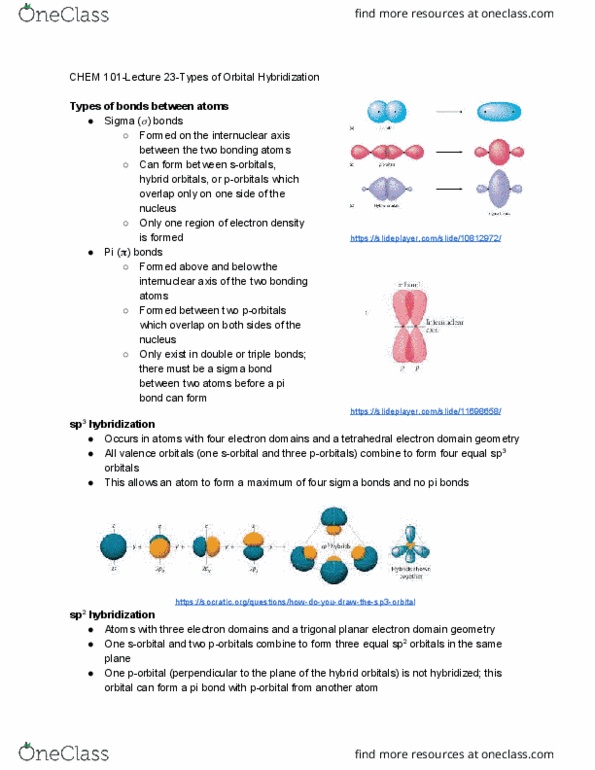
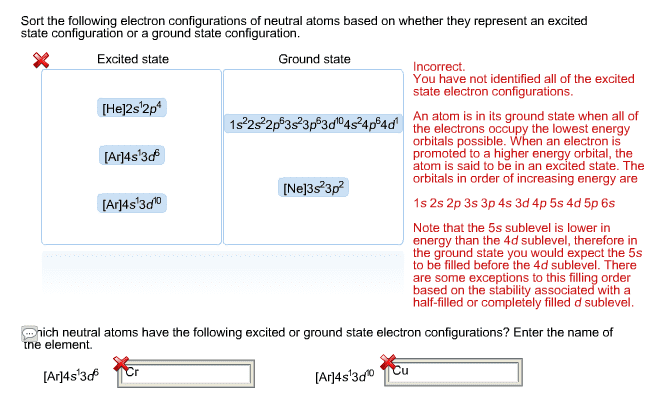
CHEM 101 Lecture Notes - Lecture 22: Pauli Exclusion Principle, Valence Electron, Vsepr Theory
CHEM 101 verified notes
22/40View all
Document Summary
Aufbau principle: fill orbitals in order of increasing energy. Exact order can be determined from the periodic table: 1s, 2s, 2p, 3s, 3p, 4s, 3d, Pauli exclusion principle: no two electrons can have the same set of quantum numbers. Each orbital can contain a maximum of two electrons; one with a + spin and one with a - spin. Hund"s rule: for degenerate energy levels, each orbital is half-filled before any are completely filled. Any valence electron which does not have a pair (it is the only electron in its orbital) can participate in bonding. In order to form more bonds, valence electrons from filled orbitals may be excited into a higher-energy orbital. The electrons which participate in bonding may be in differently shaped orbitals (s, p, d, or f), which would cause unequal angles between different bonds. According to vsepr theory, all bonds formed in a molecular geometry are equal.