CHEM 101 Lecture Notes - Lecture 10: Alkali Metal, Electron Configuration, Periodic Table
CHEM 101 verified notes
10/40View all
Document Summary
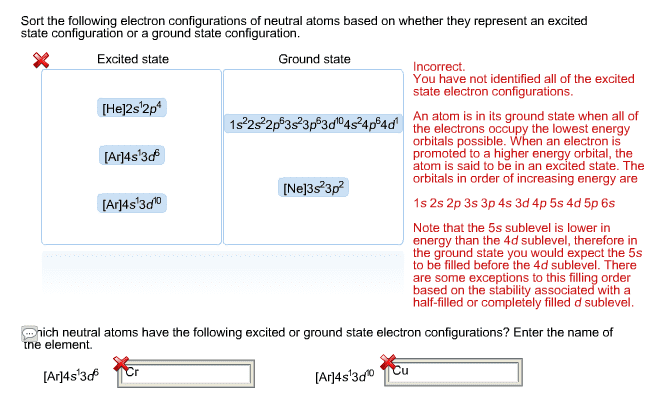
Columns are called groups , while rows are called periods. The order that the orbitals are filled in any atom, according to the aufbau principle, is: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f. This order is represented on the periodic table. Different areas on the periodic table represent different orbitals. The electron configuration of any atom can be written by listing each subshell and the number of electrons in that subshell. For atoms with many electrons, this notation can be very long. A condensed electron configuration can also be used. Focuses on the valence electrons (the electrons in the outer shell) Valence electrons participate in chemical reactions, while core electrons (electrons in inner shells) do not. The core electrons correspond to the electrons of a noble gas. Ex: for aluminum, the core electrons are 1s 2 2s 2 2p 6 , corresponding to the electrons in neon.