CHM135H1 Lecture Notes - Lecture 9: Kinetic Theory Of Gases, Molar Mass, Btg Pactual
CHM135H1 verified notes
9/38View all
Document Summary
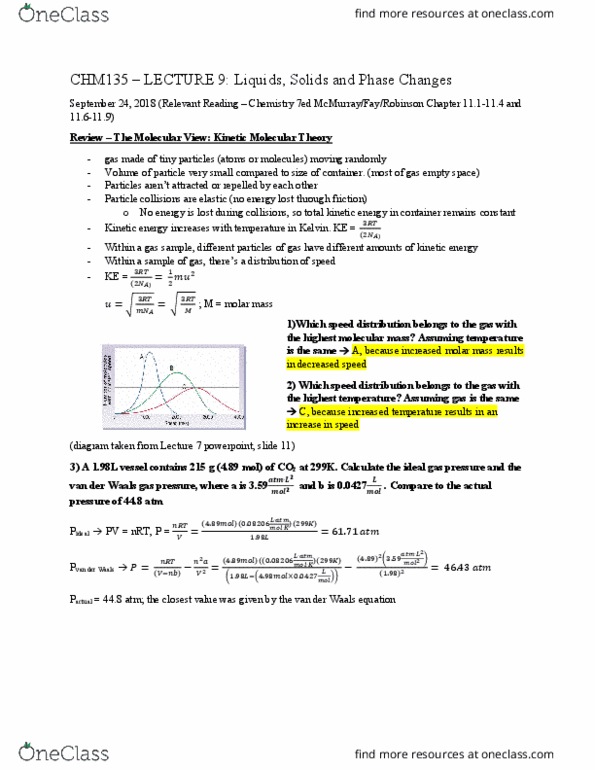
Chm135 lecture 9: liquids, solids and phase changes. September 24, 2018 (relevant reading chemistry 7ed mcmurray/fay/robinson chapter 11. 1-11. 4 and. Review the molecular view: kinetic molecular theory: no energy is lost during collisions, so total kinetic energy in container remains constant gas made of tiny particles (atoms or molecules) moving randomly. Volume of particle very small compared to size of container. (most of gas empty space) Particles aren"t attracted or repelled by each other. Particle collisions are elastic (no energy lost through friction) Within a gas sample, different particles of gas have different amounts of kinetic energy. Within a sample of gas, there"s a distribution of speed. Kinetic energy increases with temperature in kelvin. Ke = (cid:2871) (cid:1873)= (cid:2871)(cid:3040)(cid:3015)= (cid:2871)(cid:3014) ; m = molar mass. C, because increased temperature results in an increase in speed (diagram taken from lecture 7 powerpoint, slide 11: a 1. 98l vessel contains 215 g (4. 89 mol) of co2 at 299k.