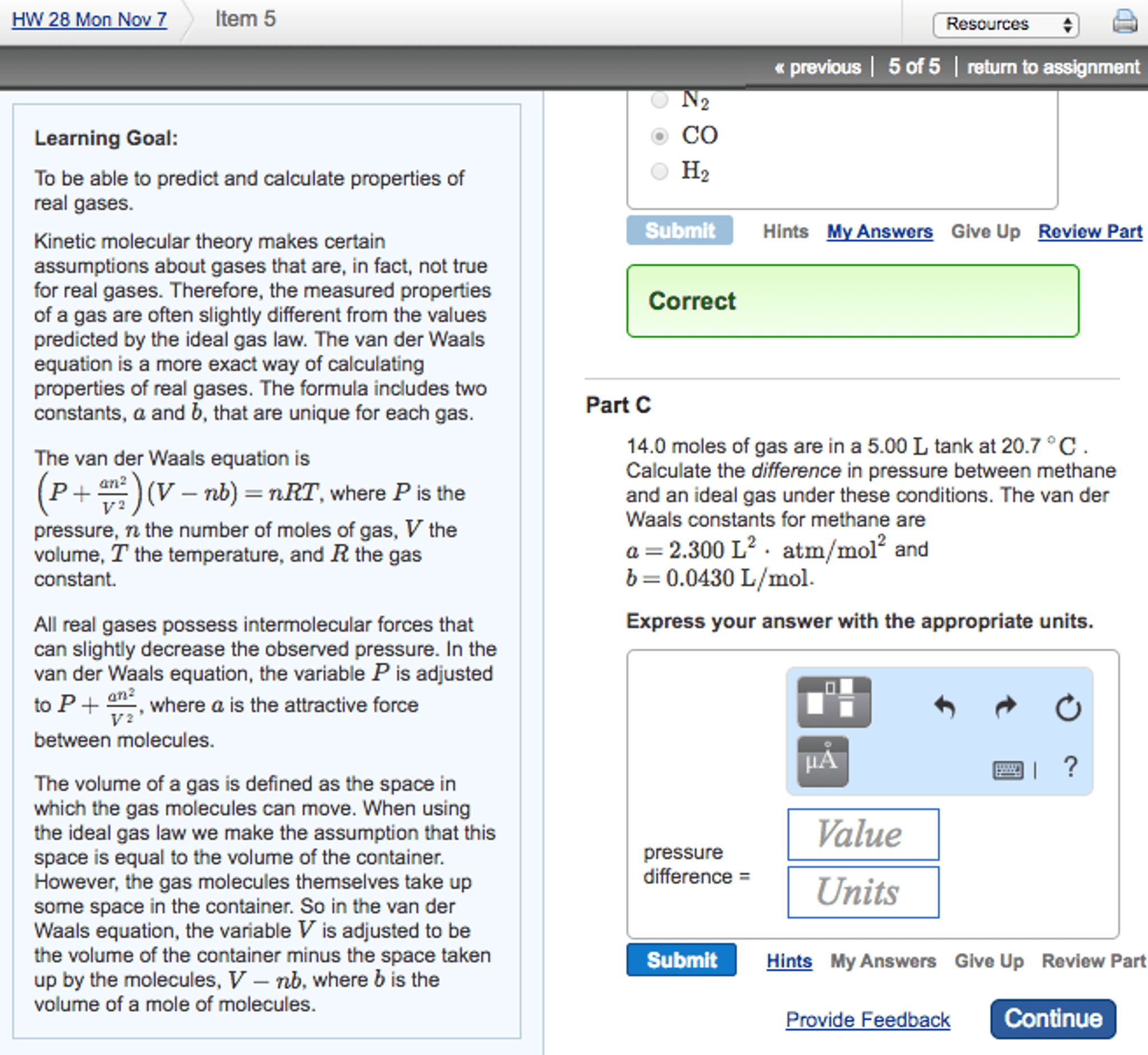
CHM135H1 Lecture Notes - Lecture 9: Van Der Waals Equation, Ideal Gas Law, Capillary Action

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
Kinetic energy increases with temperature (ke = 3 * r * t / (2n) Results from gas particles colliding with container walls. More particles -> more collisions -> greater pressure. Higher temp -> higher ke of particles -> more collisions -> greater pressure. Less volume -> more collisions -> greater pressure. High pressure -> pv/nrt > 1 due to molecular volume. Space between molecules smaller, volume of molecules becomes significant. Corrections to ideal gas law (p+n2a/v2)*(v-nb) = nrt. A -> atm * l^2 / mol ^2. Examples: 1l of gas a at 1 atm and 1l of gas b at 1atm are mixed in 2l container. Pressure of mixture is less than 1 atm: this means that molecules of b are attracted to molecules of a more than to themselves. Particles close together, but moving past each other. Surface tension : amount of energy required to expand a liquid surface.