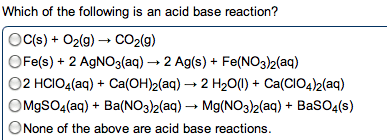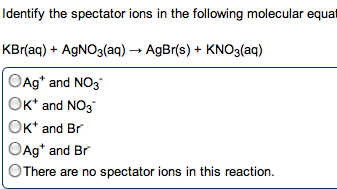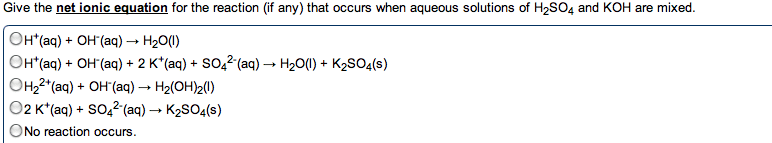CHEM 1R03 Lecture Notes - Lecture 19: Ionic Compound, Nonmetal, Sulfuric Acid

Chemistry Lecture Nineteen (short)
Types of Reactions
•When acids dissociate in water they release H+ cations and their anions
•When bases dissociate in water they release OH- anions and their cations
•In the reaction of an acid with a base, the H+ from the acid combines with the OH-
from the base to make water
•The cation from the base combines with the anion from the acid to make salt
•In general remember: acid + base --> salt + water
•For example: H2SO4 (aq) + Ca(OH)2 (aq) --> CaSO4 (aq) + 2H2O (l)
•The net equation for an acid-base reaction is always: H+ (aq) + OH- (aq) --> H2O (l)
•Next there is oxidation-reduction which is all reactions that involve a transfer of one
or more electron that is called an oxidation-reduction reactions
•We say that the substance that loses electrons in the reaction is oxidized and the
substance that gains electrons in the reaction is reduced
•This type of reaction usually occurs when a metal reacts with a non-metal
•When O2 is used as a reactant, this is called oxidizing
•For example, taking a fuel and burning it in the air basically means you are oxidizing
it
•When metals and non-metals react, the metal loses electrons and becomes a cation
which again is called oxidation
•The non-metal gains electrons and therefore becomes an anion which is called a
reduction
•In the reaction, electrons are transferred from the metal to the non-metal forming an
ionic compound
•Remember that ionic compounds are always solids unless they are dissolved in
water
•To predict the direct synthesis of a metal and a non-metal you first determine the
charges of the cation and anion
•Then balance the formulas and finally balance the equation