1
answer
0
watching
197
views
13 Dec 2019
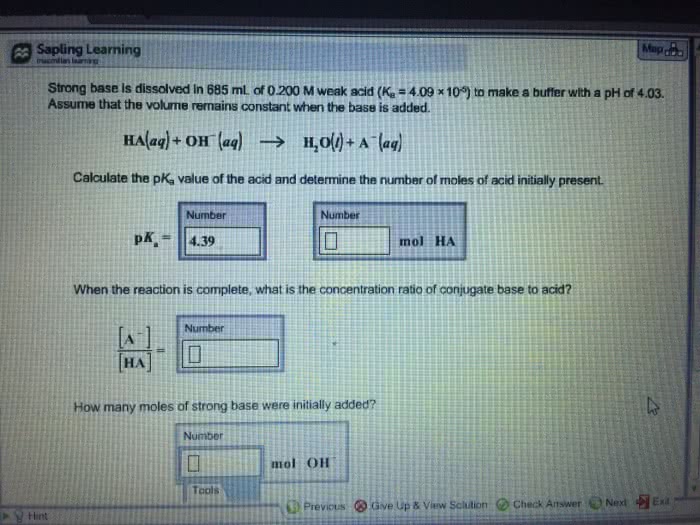
Strong base is dissolved in 715 mL of 0.400 M weak acid (Ka = 4.38 Ã 10-5) to make a buffer with a pH of 4.01. Assume that the volume remains constant when the base is added. HA(aq) + OH- (aq) -->H2O(l) + A-(aq)
A.) Strong base is dissolved in 715 mL of 0.400 M weak acid (Ka = 4.38 Ã 10-5) to make a buffer with a pH of 4.01. Assume that the volume remains constant when the base is added. HA(aq) + OH- (aq) -->H2O(l) + A-(aq)
A. )I found the PKA, which is 4.36 and the mol of HA is .286.
B.)When the reaction is complete, what is the concentration ratio of conjugate base to acid? [A-]/[HA]=?
C.)How many moles of strong base were initially added? moL[OH-]= ?
Strong base is dissolved in 715 mL of 0.400 M weak acid (Ka = 4.38 Ã 10-5) to make a buffer with a pH of 4.01. Assume that the volume remains constant when the base is added. HA(aq) + OH- (aq) -->H2O(l) + A-(aq)
A.) Strong base is dissolved in 715 mL of 0.400 M weak acid (Ka = 4.38 Ã 10-5) to make a buffer with a pH of 4.01. Assume that the volume remains constant when the base is added. HA(aq) + OH- (aq) -->H2O(l) + A-(aq)
A. )I found the PKA, which is 4.36 and the mol of HA is .286.
B.)When the reaction is complete, what is the concentration ratio of conjugate base to acid? [A-]/[HA]=?
C.)How many moles of strong base were initially added? moL[OH-]= ?
Elin HesselLv2
17 Dec 2019