CHEM 2444 Chapter Notes - Chapter 13: Steric Effects, Vinyl Chloride, Sodium Borohydride
Document Summary
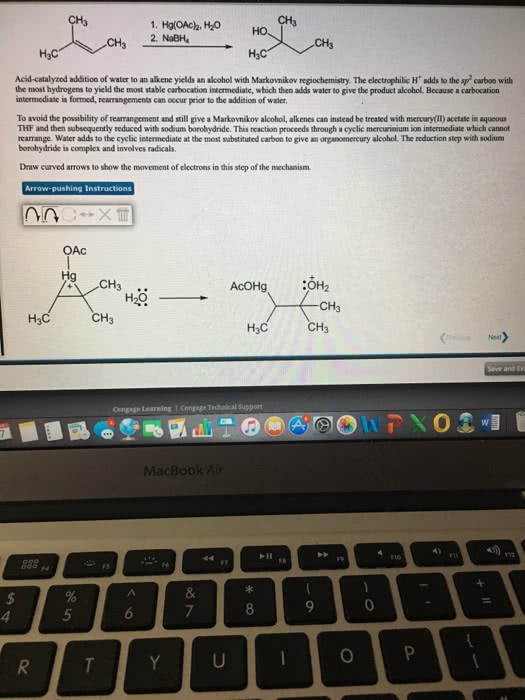
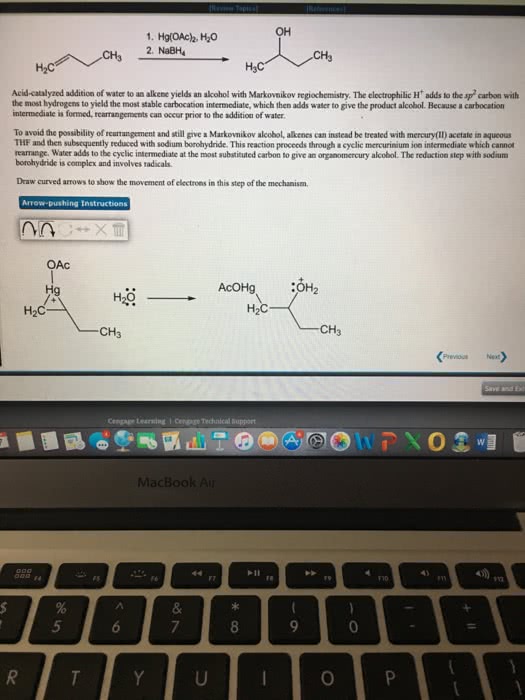
Or is always on the more highly substituted carbon because you always form the more highly substituted carbocation product: oxymercuration. Akene reacts as a lewis base, donating electrons to mercury, creating a carbon-mercury bond and breaking one of the oxygen-mercury bonds, giving a carbocation (more stable carbocation) If you do this reaction in the presence of water, the water attacks the carbocation, forms an oxonium ion, ending up with an alcohol product. Sodium borohydride (nabh4) reducing agent, introduced to mercuric alcohol product to get the markovnikov alcohol by replacing the mercury with a hydrogen. Mercury stabilizes carbocation; prevents rearrangement: hydroboration. Reacts via 4-center transition state that places b on the carbon with the least steric hindrance. In the second chemical step, borane is treated with sodium hydroxide and hydrogen peroxide to generate hydrogen peroxide anion. Boron goes on less-substituted carbon to minimize steric hindrance in the 4- centered-transition state. Enol goes under tautomerization to generate a ketone.