CHEM 1312 Study Guide - Final Guide: Lewis Acids And Bases, Lithium Hydroxide, Titration
76 views9 pages
2 Mar 2018
School
Department
Course
Professor
Document Summary
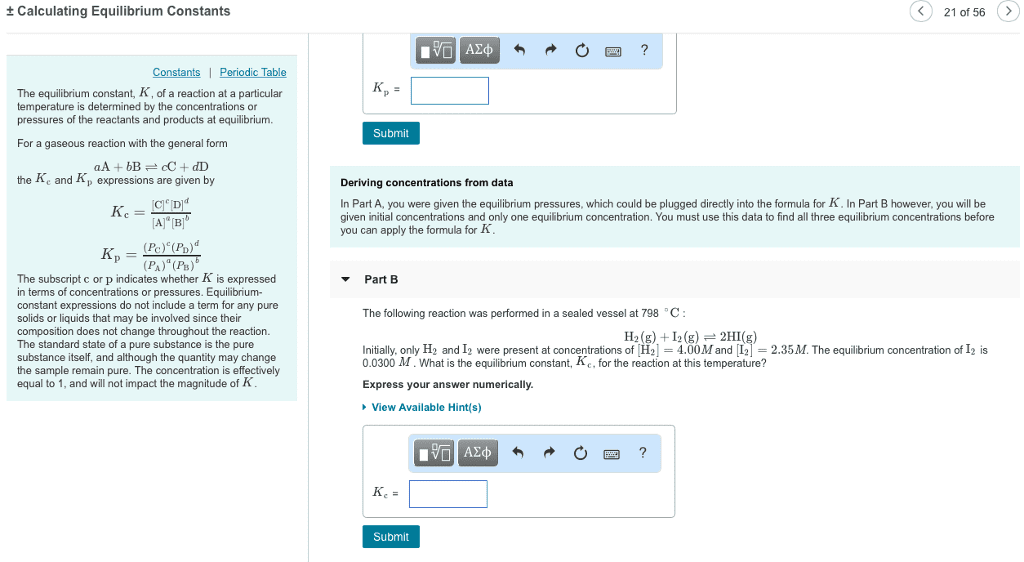
Chapter 16: chemical equilibrium equilibrium: state where the forward and reverse reactions or processes occur at the same rate. Know that concentrations are not changing at equilibrium, but they need not be equal to one another. Be able to indicate when equilibrium is achieved given concentration vs. time plots. Use the law of mass action to write equilibrium expressions for kc or kp for homogeneous and heterogeneous reactions. Include only gases; omit pure liquids and solids. For general reaction, j a + k b l c + m d. For large values of kc or kp (>103), the reaction essentially goes to completion. The equilibrium mixture consists mostly of products (product favored) For small values of kc or kp (<10-3), the reaction does not occur to any significant degree. The equilibrium mixture consists mostly of reactants (reactant favored) For intermediate values (10-3 the equilibrium mixture contains appreciable amounts of both reactants and products.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232