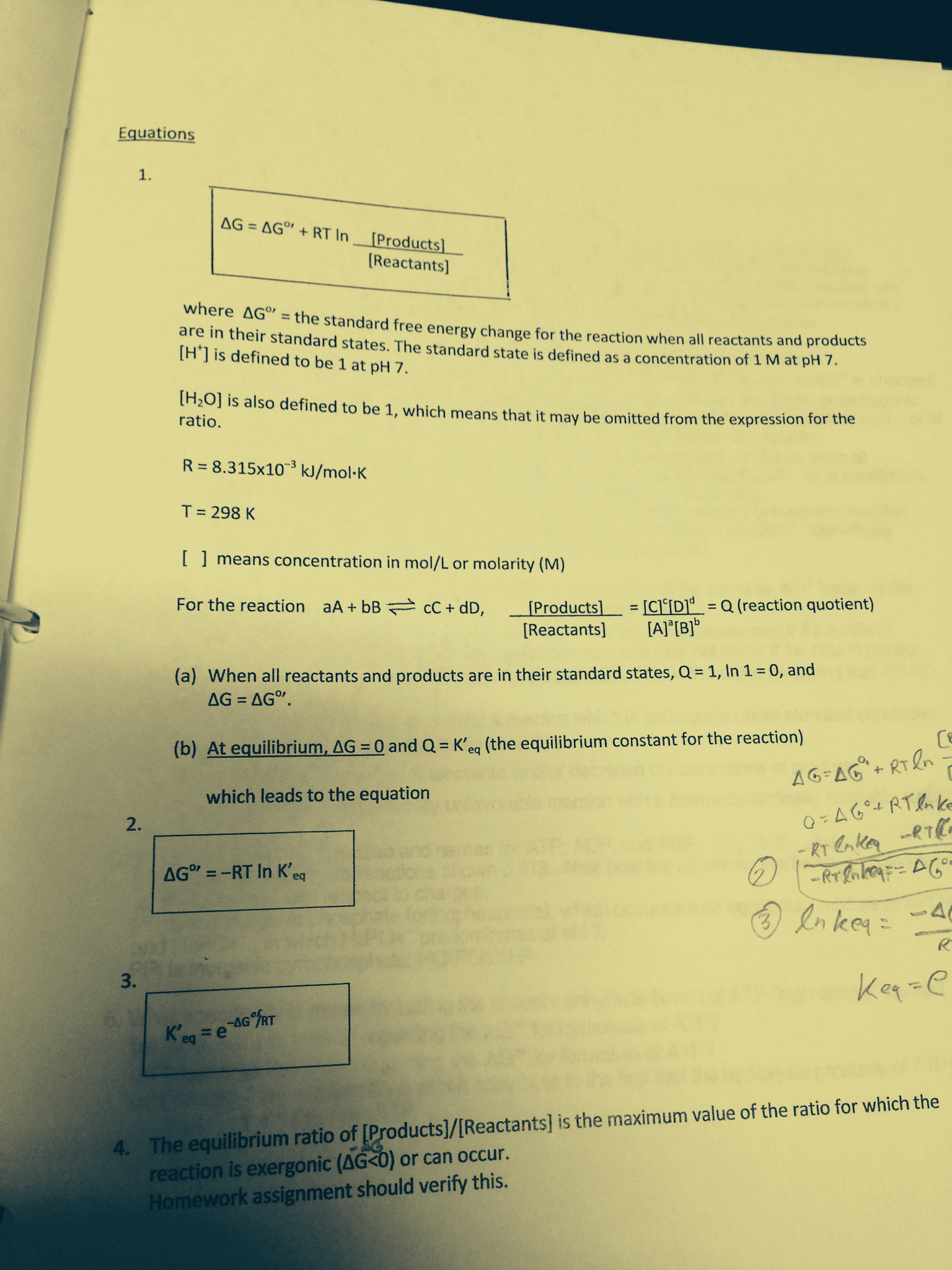CHEM 1312 Study Guide - Midterm Guide: Atomic Radius, Conjugate Acid, Endothermic Process
46 views7 pages
1 Mar 2018
School
Department
Course
Professor
Document Summary
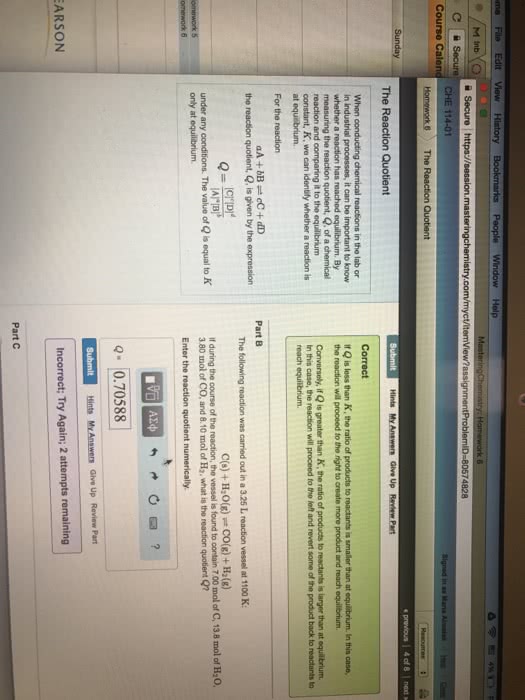
Please note that there might be errors in this document. The variables equation by placing brackets around a given product or reactant. (cid:2185),(cid:2198),(cid:2173), and equilibrium (cid:3030) is defined as the concentration equilibrium constant. It can be denoted in an (cid:3043) is defined as the (partial) pressure equilibrium constant. It can be denoted in an (cid:1843)(cid:3030) and (cid:1843)(cid:3043) are the reaction quotients of their indicated notations. Reaction constant equation by placing a p in front of a given product or reactant. [(cid:1840)(cid:1841)(cid:2871) ] (cid:1842)(cid:3015)(cid:3016)(cid:3119) may apply to a reaction any time that the reaction is not in equilibrium. Reactants are not required to equal products in equilibrium, however they may. If k is larger than one, it has more products than reactants. If k is smaller than one, it has fewer products than reactants. If k is one, the products and reactants are equal.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232