[BCHM 3010] - Midterm Exam Guide - Ultimate 28 pages long Study Guide!
Document Summary
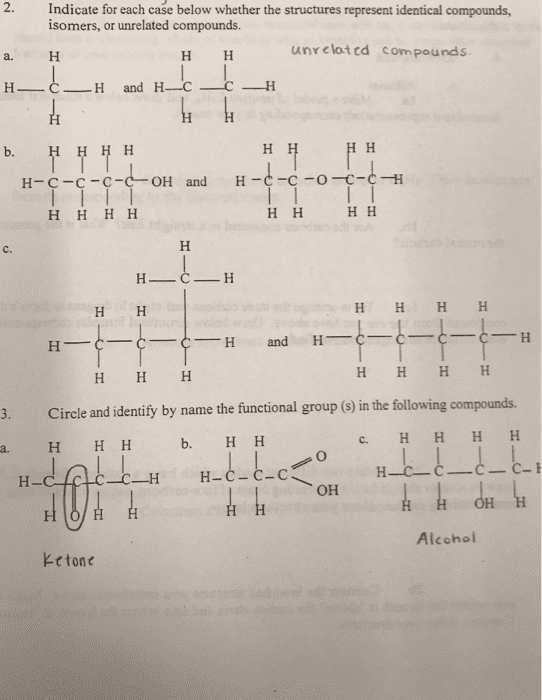
Chapter 1 - textbook problems: 7: in each of the compounds below, circle and identify by name each functional group. A strong acid will dissociate completely and the h+ in the equation will increase with acid strength. As the numerator increases the ka also increases. The compound which has the tendency to accept a proton is known as a. At ph 1. 0, it would be fully protonated. ph 6. 2 is close to the pi of. Alanine (6. 2) therefore it would be in zwitterion form. ph 8. 02 zwitterion. ph 11. 9 fully deprotonated. Clockwise rotation is known as dextrorotatory, counterclockwise is known as levorotatory. (clockwise should spell corn for l, conr for d) Tryptophan and tyrosine absorb ultraviolet light (much lesser extent phenylalanine) because they are aromatic. Amphoteric (ampholytes) - can act as either acid or base because they are zwitterions. At low ph (protonated) and at high ph (deprotonated). Henderson-hasselbach equation: ph = pk + log ([a-]/[ha]) pi = (pk1+pk2) (1/2)