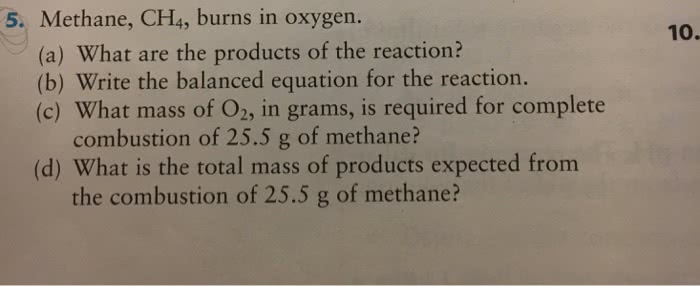14
answers
2
watching
123
views
1 Mar 2023
What mass of water (in grams) can be obtained from the complete combustion of 50.0 g of methane according to this balanced equation? CH4 (g) + 2O2 (g) → CO2 (g) + 2H2O (g)
What mass of water (in grams) can be obtained from the complete combustion of 50.0 g of methane according to this balanced equation? CH4 (g) + 2O2 (g) → CO2 (g) + 2H2O (g)
wahabmunir796Lv10
12 Jul 2023
5 Mar 2023
Already have an account? Log in
nicolaidoscopeLv10
2 Mar 2023
Already have an account? Log in
1 Mar 2023
Already have an account? Log in
davidinoooLv7
1 Mar 2023
Already have an account? Log in
redsunvuongLv3
1 Mar 2023
Already have an account? Log in
celluarsolLv10
1 Mar 2023
Already have an account? Log in