1
answer
0
watching
12
views
11 Dec 2019
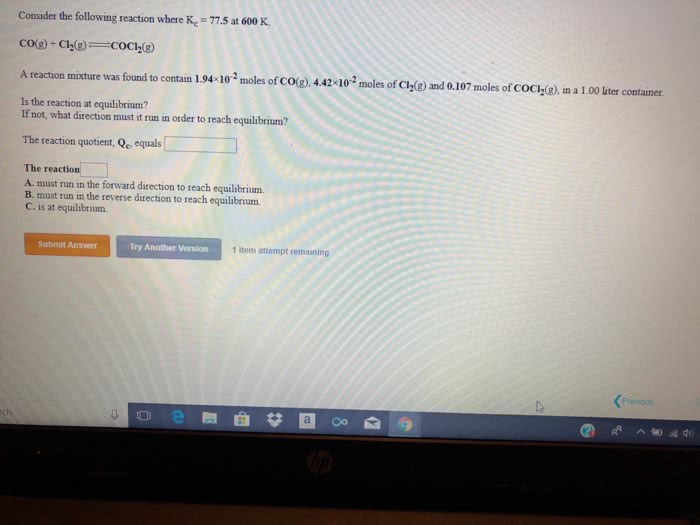
Consider the following reaction where Kc = 55.6 at 698 K.
H2(g) + I2(g) 2HI(g)
A reaction mixture was found to contain 2.54Ã10-2 moles of H2(g), 4.07Ã10-2 moles of I2(g) and 0.294 moles of HI(g), in a 1.00 liter container.
Is the reaction at equilibrium?
If not, what direction must it run in order to reach equilibrium?
The reaction quotient, Qc, equals ________?
The reaction: ________
A. must run in the forward direction to reach equilibrium.
B. must run in the reverse direction to reach equilibrium.
C. is at equilibrium.
Consider the following reaction where Kc = 55.6 at 698 K.
H2(g) + I2(g) 2HI(g)
A reaction mixture was found to contain 2.54Ã10-2 moles of H2(g), 4.07Ã10-2 moles of I2(g) and 0.294 moles of HI(g), in a 1.00 liter container.
Is the reaction at equilibrium?
If not, what direction must it run in order to reach equilibrium?
The reaction quotient, Qc, equals ________?
The reaction: ________
A. must run in the forward direction to reach equilibrium.
B. must run in the reverse direction to reach equilibrium.
C. is at equilibrium.
george-123Lv2
23 Mar 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in