2
answers
0
watching
201
views
11 Nov 2019
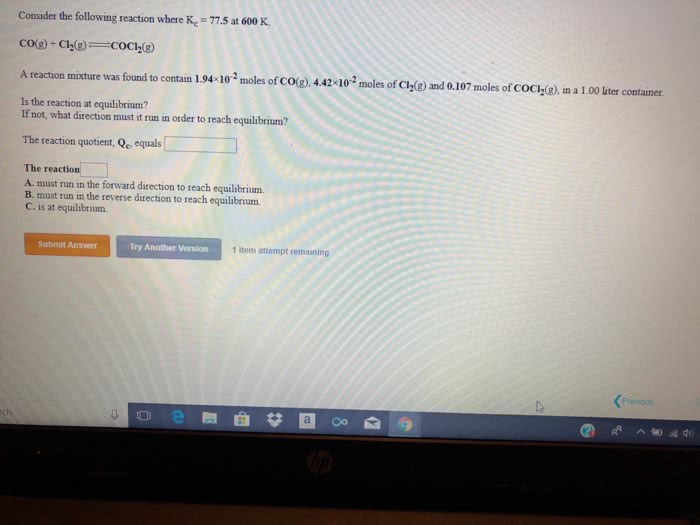
Consider the following reaction where Kc = 77.5 at 600 K.
CO(g) + Cl2(g) <---->COCl2(g)
A reaction mixture was found to contain 2.42Ã10-2 moles of CO(g), 3.94Ã10-2 moles of Cl2(g) and 0.116 moles of COCl2(g), in a 1.00 liter container.
Is the reaction at equilibrium?
If not, what direction must it run in order to reach equilibrium?
The reaction quotient, Qc, equals________ .
The reaction________
A. must run in the forward direction to reach equilibrium.
B. must run in the reverse direction to reach equilibrium.
C. is at equilibrium.
Consider the following reaction where Kc = 77.5 at 600 K.
CO(g) + Cl2(g) <---->COCl2(g)
A reaction mixture was found to contain 2.42Ã10-2 moles of CO(g), 3.94Ã10-2 moles of Cl2(g) and 0.116 moles of COCl2(g), in a 1.00 liter container.
Is the reaction at equilibrium?
If not, what direction must it run in order to reach equilibrium?
The reaction quotient, Qc, equals________ .
The reaction________
A. must run in the forward direction to reach equilibrium.
B. must run in the reverse direction to reach equilibrium.
C. is at equilibrium.
george-123Lv2
23 Mar 2023
Keith LeannonLv2
3 Jul 2019
Already have an account? Log in