1
answer
0
watching
317
views
12 Dec 2019
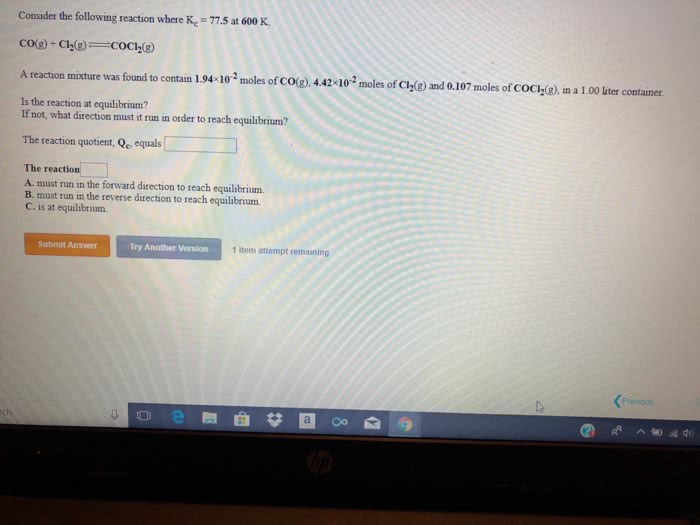
Consider the following reaction where Kc=1.20x10-2 at 500K.
PCl5(g) PCl3(g)+Cl2(g)
A reaction mixture was found to contain 0.117 moles of PCl5(g), 4.15x10-2 moles of PCl3(g), and 4.51x10-2 moles of Cl2(g), in a 1.00 liter container.
Is the reaction at equilibrium?
If not,what direction must it run in order to reach equilibrium?
The reaction quotient, Qc, equals
The reaction:
A. must run in the forward direction to reach equilibrium.
B. must run in the reverse direction to reach equilibrium.
C. is at equilibrium
Consider the following reaction where Kc=1.20x10-2 at 500K.
PCl5(g) PCl3(g)+Cl2(g)
A reaction mixture was found to contain 0.117 moles of PCl5(g), 4.15x10-2 moles of PCl3(g), and 4.51x10-2 moles of Cl2(g), in a 1.00 liter container.
Is the reaction at equilibrium?
If not,what direction must it run in order to reach equilibrium?
The reaction quotient, Qc, equals
The reaction:
A. must run in the forward direction to reach equilibrium.
B. must run in the reverse direction to reach equilibrium.
C. is at equilibrium
Hubert KochLv2
13 Dec 2019