1
answer
0
watching
230
views
18 Nov 2019
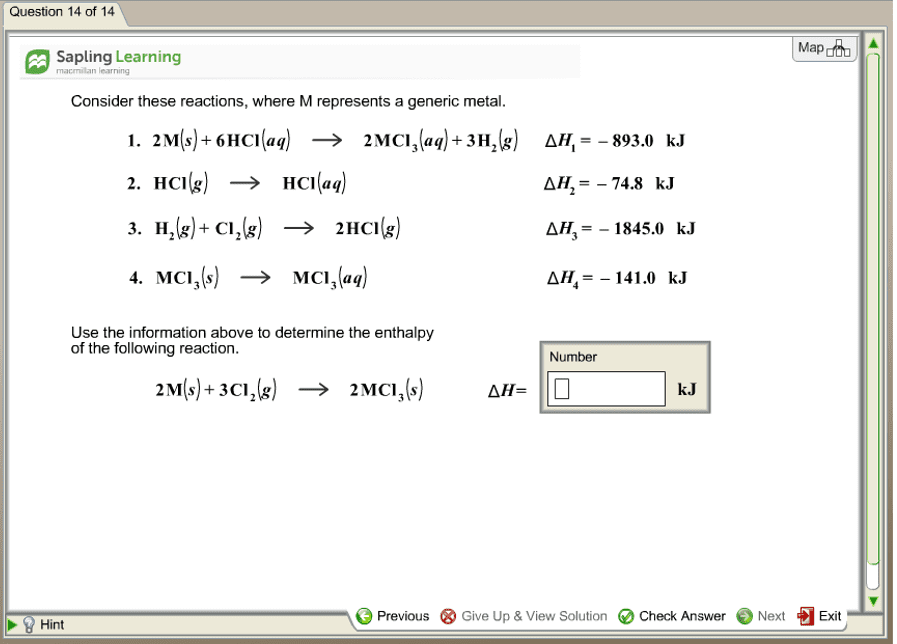
Consider these reactions, where M represents a generic metal. 2M(s) + 6HCl(aq) rightarrow 2 MCl_3(aq) + 3H_2(g) delta H_1 = - 893.0 kJ HCl(g) rightarrow HCl(aq) delta H_2 = - 74.8 kj H_2(g) + Cl_2(g) rightarrow 2HCl(g) delta H_3 = - 1845.0 kJ MCl_3(s) rightarrow MCl_3(aq) delta H_4 = - 141.0 kj Use the information above to determine the enthalpy of the following reaction. 2M(s) + 3Cl_2(g) rightarrow 2MCl_3(s) delta H =

Consider these reactions, where M represents a generic metal. 2M(s) + 6HCl(aq) rightarrow 2 MCl_3(aq) + 3H_2(g) delta H_1 = - 893.0 kJ HCl(g) rightarrow HCl(aq) delta H_2 = - 74.8 kj H_2(g) + Cl_2(g) rightarrow 2HCl(g) delta H_3 = - 1845.0 kJ MCl_3(s) rightarrow MCl_3(aq) delta H_4 = - 141.0 kj Use the information above to determine the enthalpy of the following reaction. 2M(s) + 3Cl_2(g) rightarrow 2MCl_3(s) delta H =
Jamar FerryLv2
28 Oct 2019

