1
answer
0
watching
367
views
6 Nov 2019
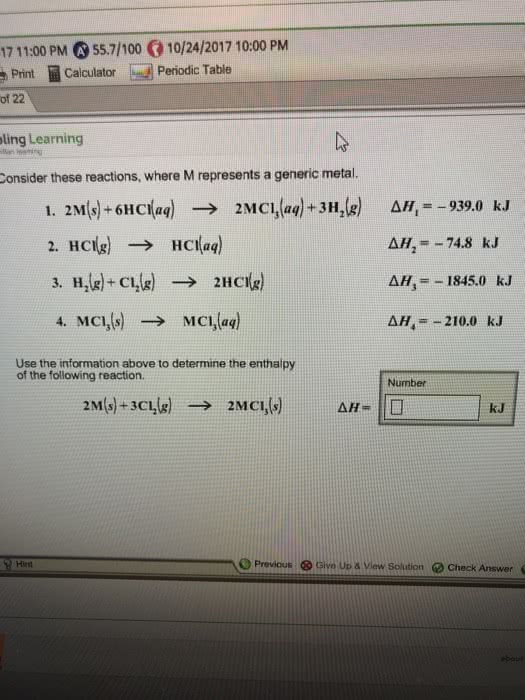
Consider these reactions, where M represents a generic metal.
2M(s)+6HCl(aq)â¶2MCl3(aq)+3H2(g) Îð»1=â856.0 kJ
HCl(g)â¶HCl(aq) Îð»2=â74.8 kJ
H2(g)+Cl2(g)â¶2HCl(g) Îð»3=â1845.0 kJ
MCl3(s)â¶MCl3(aq) Îð»4=â437.0 kJ
Use the given information to determine the enthalpy of the reaction
2M(s)+3Cl2(g)â¶2MCl3(s)
Îð»= _____kJ
Consider these reactions, where M represents a generic metal.
2M(s)+6HCl(aq)â¶2MCl3(aq)+3H2(g) Îð»1=â856.0 kJ
HCl(g)â¶HCl(aq) Îð»2=â74.8 kJ
H2(g)+Cl2(g)â¶2HCl(g) Îð»3=â1845.0 kJ
MCl3(s)â¶MCl3(aq) Îð»4=â437.0 kJ
Use the given information to determine the enthalpy of the reaction
2M(s)+3Cl2(g)â¶2MCl3(s)
Îð»= _____kJ
Hubert KochLv2
7 May 2019