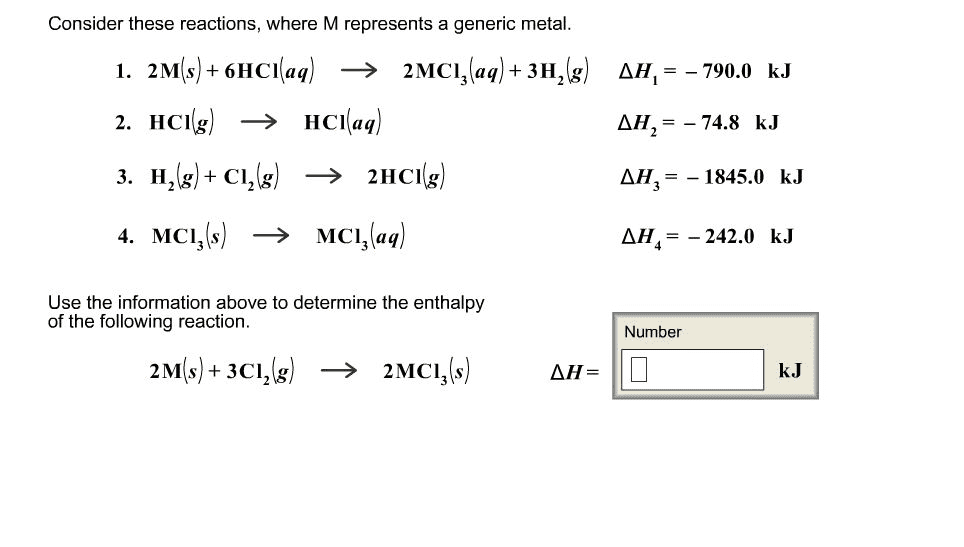1
answer
0
watching
2,506
views
11 Dec 2019
Consider these reactions where M represents a generic metal:
2M(s) + 6HCl(aq) ---->2MCl3(aq) + 3H2(g) Delta H1 = -569kj
HCl(g) ----> HCl(aq) DeltaH2 = -74.8
H2(g) + Cl2(g) ----> 2HCl(g) Delta H3 = -1845kj
MCl3(s) ----> MCl3(aq) Delta H4 = -405kj
Use the above information to determine the enthalpy of the following reaction:
2M(s) + 3Cl2(g) ----> 2MCl3(s) What is Delta H for this reaction?
Consider these reactions where M represents a generic metal:
2M(s) + 6HCl(aq) ---->2MCl3(aq) + 3H2(g) Delta H1 = -569kj
HCl(g) ----> HCl(aq) DeltaH2 = -74.8
H2(g) + Cl2(g) ----> 2HCl(g) Delta H3 = -1845kj
MCl3(s) ----> MCl3(aq) Delta H4 = -405kj
Use the above information to determine the enthalpy of the following reaction:
2M(s) + 3Cl2(g) ----> 2MCl3(s) What is Delta H for this reaction?
Verified Answer
Tod ThielLv2
13 Dec 2019
Joey Tang
Bachelor’s Degree in Chemistry from McMaster University14 Apr 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.