CHEM 2323 Study Guide - Alkyne, Stereochemistry, Acetylide
Document Summary
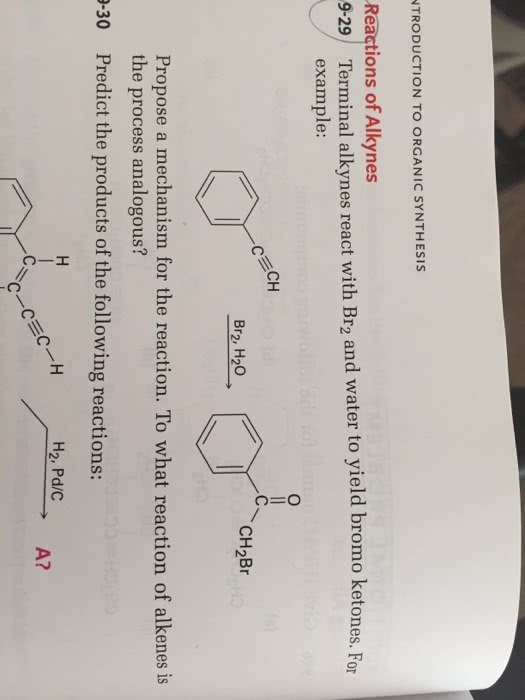
Chapter 9 reactions deal with synthesis and reactions of alkynes. For the bare minimum, memorize what reagent is used, and what product is formed. For alkynes, you don"t have to memorize the mechanisms, but again it helps to at least understand it to make it easier to predict the products. I will try to emphasize which reactions are important, and what you should know for each reaction shown below. A terminal alkyne hydrogen is relatively acidic (pka ~25), and can become deprotonated using a strong base (nh2 in a sn2 reaction with a common electrophile (must be a primary, unhindered, electrophile!). (you don"t need to specify stereochemistry) The alkyne ion can then react as a good nucleophile. If reacting with an acetylide, specify 1 equivalent so you only extract 1 hydrogen. Similar to the previous reaction, nanh2 can extract a hydrogen from a terminal alkyne and form a good nucleophile.