CHEM 1A Lecture 26: 11-30 Lecture 26

94
CHEM 1A Full Course Notes
Verified Note
94 documents
Document Summary
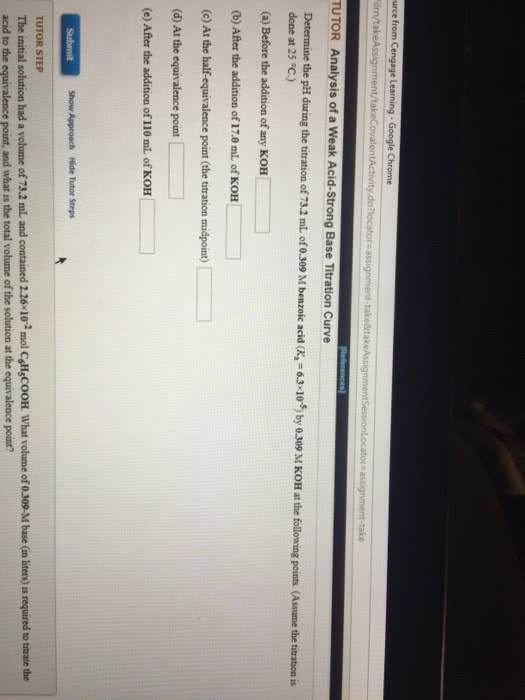
Adding a strong base allows to find the ph. When you add a strong entity into the buffer system, convert it to a weak. The net ionic equation will be h+ + oh- h2o. The equivalence point is the same amount of acid and base. (7) Calculated the ph before the equivalence point (random amount added as long as it"s before the equivalence point) (5 ml). Titrate 25. 0ml of 1. 0 m ch3cooh with 1. 0 m naoh. Before and at the equivalence point (25. 0 ml of 1. 0 m naoh) Two step problem: 1) stoichiometry 2) equilibrium. Similar type of problem but have some nuances see example 8. 2b for before equivalence point and last problem. Major species during titration: ch3cooh, oh-, na+, h2o. Ch3cooh + oh- ch3coo- + h2o. 0. 025 mol -0. 025 mol + 0. 025 mol. Major species in solution: ch3coo-, na+, h2o. Reaction: ch3coo- + h2o ch3cooh + oh-