CHEM 1A Lecture Notes - Lecture 25: Buffer Solution, Sodium Hydroxide, Stoichiometry
18 views2 pages

94
CHEM 1A Full Course Notes
Verified Note
94 documents
Document Summary
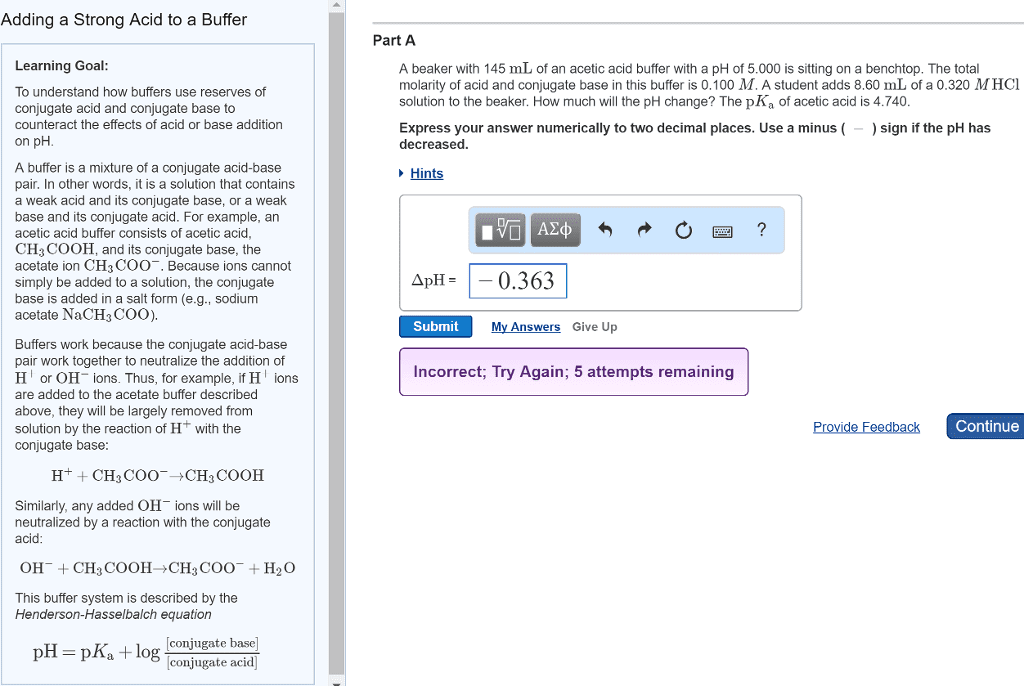
Function of a weak acid in a buffer. The function of the weak acid in a buffer is to neutralize added base. The acetate ion produced by the neutralization becomes part of the available acetate. Hc2h3o2 (aq) + oh- (aq) c2h3o2. Ch3cooh + oh- ch3coo- + h2o. Major species left in solution: oh- used up. Buffer solution: ph = 4. 75 (+ 0. 08) 40 ml di water + 2 ml 1. 0 m naoh. 40 ml buffer solution + 2 ml 1. 0 m naoh. Original buffer solution, ph add h+ or oh- two step problem modified ph. Step 1: let reaction, h+ with a- or oh- with ha, go to completion . Stoichiometry problem determine new concentrations of ha and a- Titrate a strong acid with a strong base. Strong acids or strong bases, such as h+ or oh- go to completion. Equivalence point - the point in a titration where the moles of acid = moles of base.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232