CHEM 2261 Chapter : Notes2261Chapter6

60
CHAPTER 6: IONIC REACTIONS
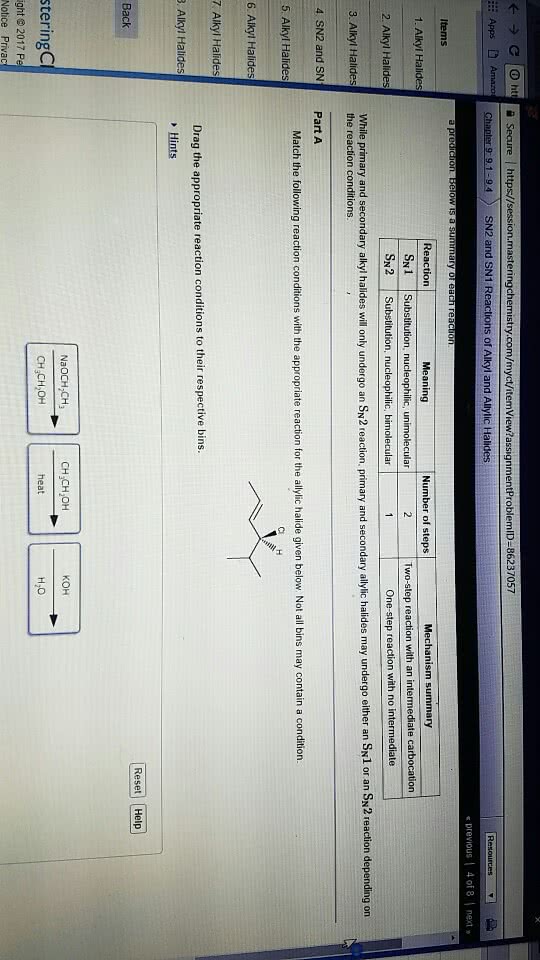
Substitution vs. elimination reaction
Compounds with an sp3 carbon bonded to a electronegative
atom or group (e.g. as in alkyl halides) can undergo 2 types of
reactions: substitution and/or elimination:
RXY
RYX
RHY X
substitution
elimination
nucleophile
alkyl
halide leaving
group
The SN2 reaction (substitution nucleophilic bimolecular)
The rate of a nucleophilic substitution reaction depends upon:
(1) the structure of the alkyl halide
(2) the leaving group
(3) the nucleophile
(4) the solvent.
Mechanism of an SN2 reaction:
C Br
CH3CH2
H3C
HC
CH2CH3
CH3
H
Br
HO CHO
CH3
H
CH2CH3
HO +
transition state
(S)-2-bromobutane (R)-2-butanol
Rate = k [alkyl halide][nucleophile]
The rate of an SN2 reaction depends on the concentration of
both the alkyl halide and the nucleophile, therefore it is a
bimolecular reaction (second order).

61
In an SN2 reaction the reactivity order of alkyl halides (due to
steric effects) is:
H
C
H
H
Methyl halide
X>
>
H
C
H
R
1o halide
H
C
R
R
2o halide
R
C
R
R
3o halide
XXX
>
>
>
>
SN2 reactions take place in a single step, without intermediates.
The nucleophile attacks the substrate from a direction 180o away
from the leaving group, causing inversion of configuration of the
molecule.
Another example:
C Br
H
H3C
H3CH2C
II C Br
H3CCH2CH3
H
CI
H
CH3
CH2CH3
S R
The leaving group in the SN2 reaction
The leaving ability of a group is a measure of its ease of
displacement.
Weak bases are good leaving groups (the weaker the basicity of
a group, the better its leaving ability)
Two trends that determine basicity:
(1) Periodic table trend, and (2) Resonance
Order of leaving abilities of halide ions: I- > Br- > Cl- > F-
The following are excellent leaving groups:
H3C S
O
O
O F3C S
O
O
O
mesylate
(Ms) triflate tosylate
(Ts)
SH3C
O
O
O

62
The nucleophile in the SN2 reaction
Nucleophilicity is a measure of how fast a nucleophile attacks
an electron-deficient atom.
What factors determine nucleophilicity?
1. Nucleophilicity increases from right to left in the periodic
table (parallels basicity) CH3- > OH- > F-
2. Nucleophilicity increases down a group in the periodic table
(inverse relation to basicity) I- > Br- > Cl- > F-
CH3SH > CH3OH
3. When the nucleophile and basic sites are the same atom the
nucleophilicity parallels basicity. OH- > H2O; NH2- > NH3
CH3O- > CH3COO-
Effect of sterics on nucleophilicity
A non-sterically hindered species is a better nucleophile than a
bulky species (because it is harder for a bulky nucleophile to
approach the substrate). CH3O- > Me3CO-
Strong Nucleophiles(SN2):I-, CN-, N3-, OH-, CH3O-, CH3CH2O-
WeakNucleophiles(SN1): H2O, CH3OH, CH3CH2OH, CH3CO2H
Effect of the solvent on the rate of SN2 reactions.
Polar aprotic solvents do not have an H bound to an
electronegative atom and are not capable of H-bonding.
CH3CN(CH3)2
O
acetone
dimethylsulfoxide
(DMSO)
N,N-dimethylformamide
(DMF)
CH3SCH3
O
CH3CCH3
O
O
tetrahydrofuran
(THF)
SN2 is typically favored by a high concentration of a good
nucleophile (e.g., negatively charged) in a polar aprotic solvent.
Document Summary
Compounds with an sp3 carbon bonded to a electronegative atom or group (e. g. as in alkyl halides) can undergo 2 types of reactions: substitution and/or elimination: The rate of a nucleophilic substitution reaction depends upon: (1) the structure of the alkyl halide (2) the leaving group (3) the nucleophile (4) the solvent. The rate of an sn2 reaction depends on the concentration of both the alkyl halide and the nucleophile, therefore it is a bimolecular reaction (second order). In an sn2 reaction the reactivity order of alkyl halides (due to steric effects) is: Sn2 reactions take place in a single step, without intermediates. The nucleophile attacks the substrate from a direction 180o away from the leaving group, causing inversion of configuration of the molecule. The leaving group in the sn2 reaction. The leaving ability of a group is a measure of its ease of displacement.