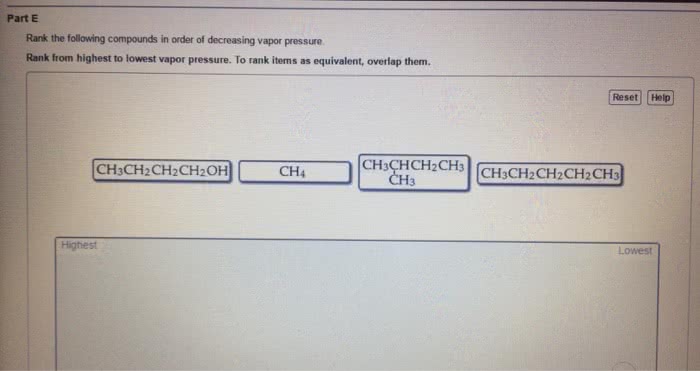
CHEM 1112 Chapter Notes - Chapter 12: Vapor Pressure, Intermolecular Force, Lattice Energy
Document Summary
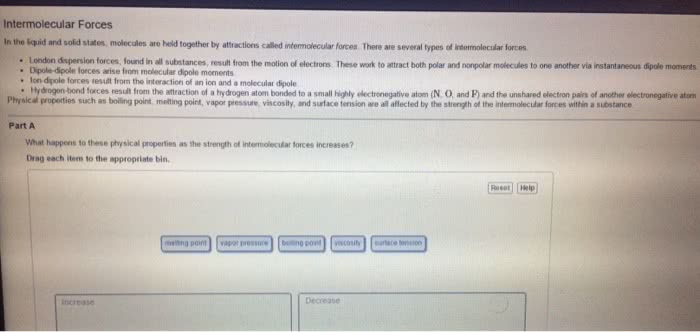
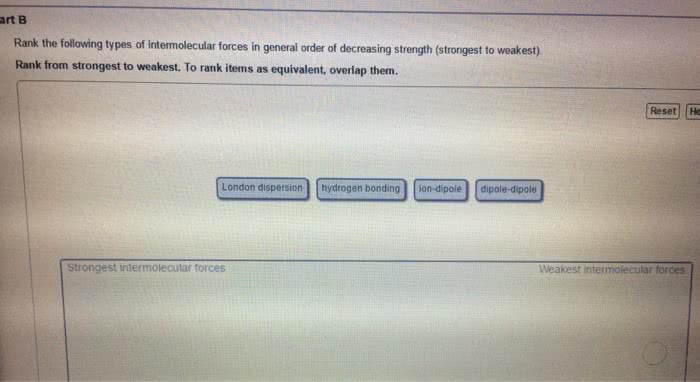
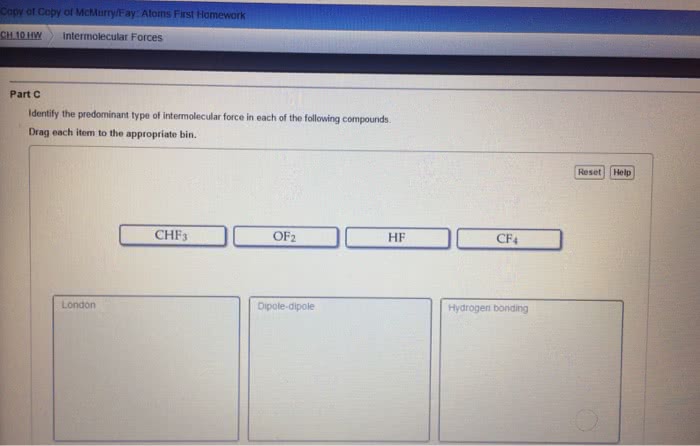
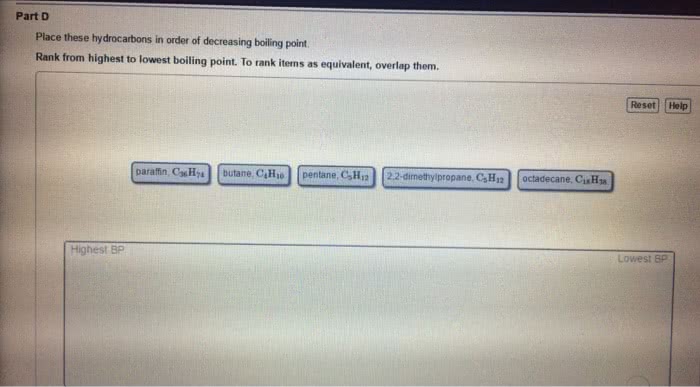
Condensed phases = liquids and solids: intermolecular forces are large enough to keep the particles of a substance in contact with one another, liquids. Substances in which attractive forces are strong enough to keep the particles together, but weak enough to allow flow (molecules moving past one another: solids. Substances in which the attractive forces are strong enough to keep the particles in fixed positions relative to one another. Molecule within liquid is pulled in all directions by intermolecular forces between it and the other molecules that surround it: no net pull in any one direction. Molecular at surface of liquid is pulled down and to the sides by neighboring molecules. There is no upward pull to balance downward/inward pull: results in a net pull inward on molecules at the surface. Causes liquid"s surface to tighten like elastic film: minimizes surface area, real-life example: beading of water on surface of freshly washed car, surface tension.