CHE-2211 Chapter Notes - Chapter 14: Thermodynamic Cycle, Stephen Hawking
Document Summary
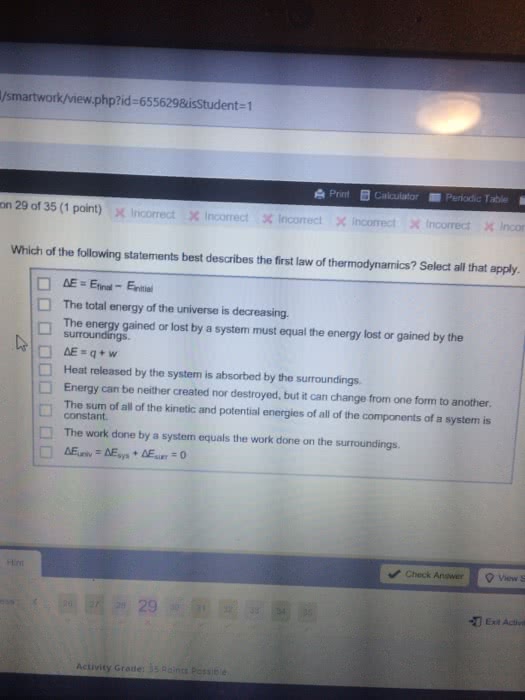
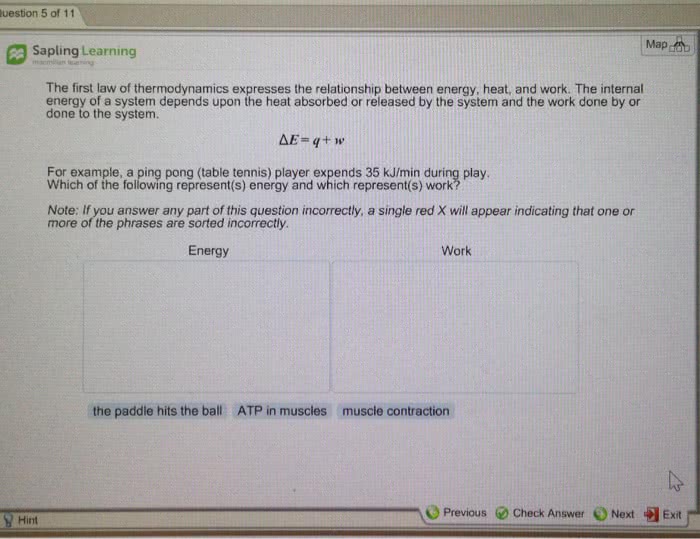
The first law of thermodynamics states that energy is not created, nor destroyed, but is conserved. Then this law expresses that, when a system is subjected to a thermodynamic cycle, the heat transferred by the system will be equal to the work received by it, and vice versa. That is, q = w, where q is the heat supplied by the system to the environment and w the work done by the environment to the system during the cycle. A simple example would be: when removing the water contained in a container with a drill, we are applying work to it, which is equal to the heat it emits to the environment when it is heated. In this case, the system can be water, the medium would be the drill, the surrounding air and everything outside the system that is not water (because what is outside will receive heat from the system).