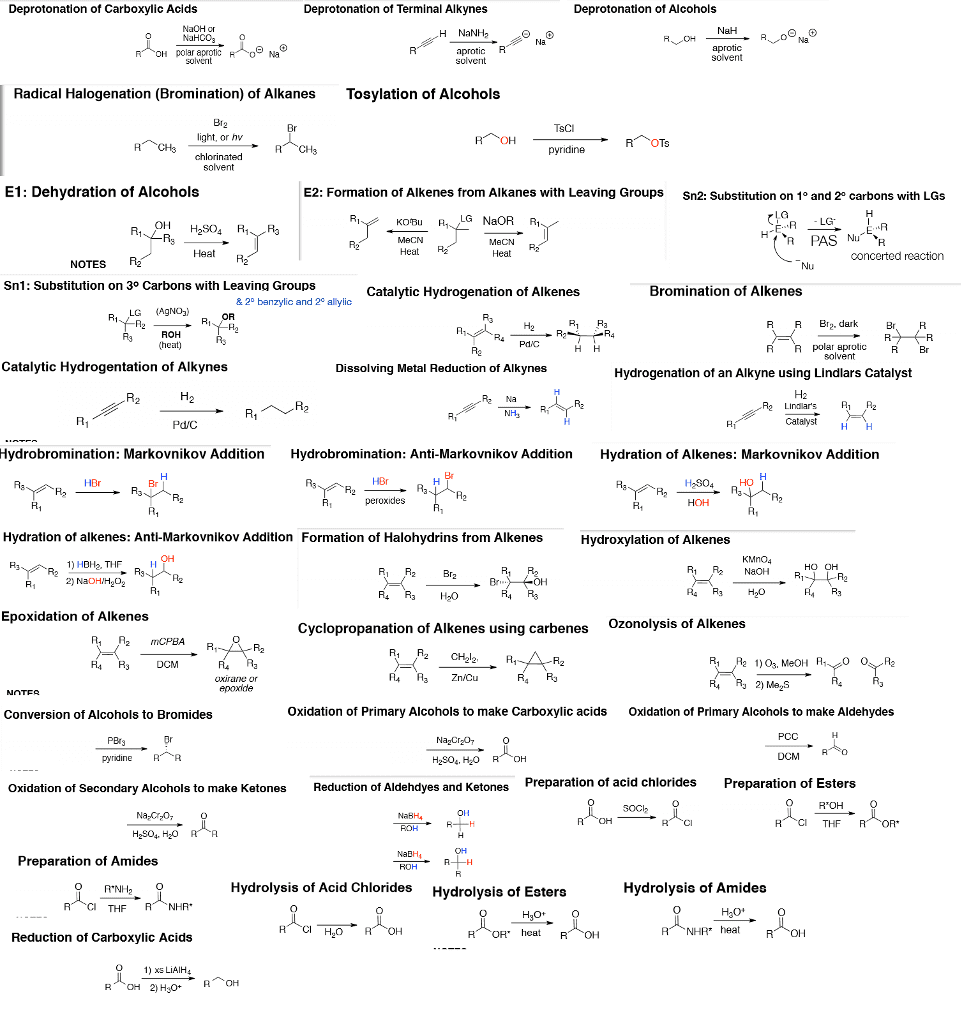
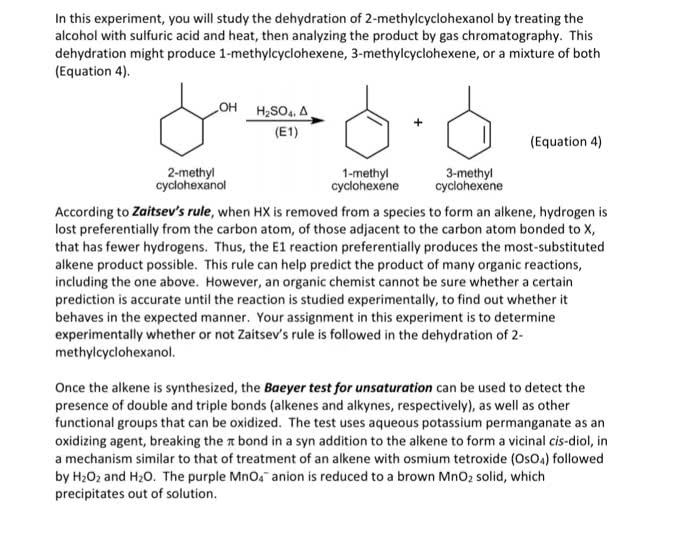
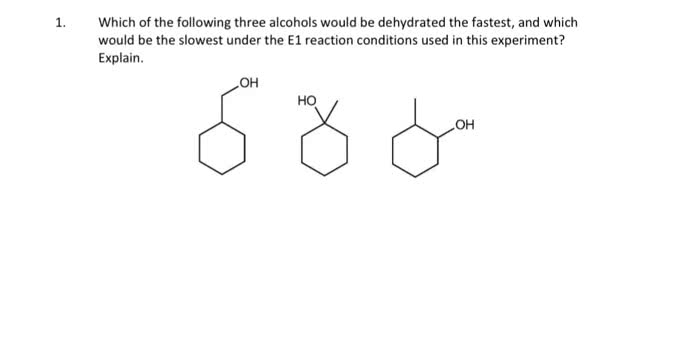
Chemistry 1027A/B Chapter Notes - Chapter 2: Steric Effects, Syn And Anti Addition, Epoxide
Document Summary
Rate dependent on concentration of r-lg and nu. 1 or 2 alkyl halides (sn2) (not 3 - elimination) Addition of o to alkene in 3 membered ring + Oh on both carbons bonded to 3- membered ring. Resonance stabilization of oxygen makes them stronger acids. Oxidation of benzylic carbons to carboxylic acids if bonded to one or more h. Fex3 + x2 x- + fex4 acts as base to remove h. Nitro group reduced to amine with 3h2 & pt, Pd, ni yields benzene ring with nh2. 2 moles amines: lone pair moderate nucleophiles. 1st mole reacts with nu to give product. 2nd mole deprotonates and then reacts with coproduct to neutralize and form ammonium salt. Ion product then converted to alcohol using h+/h2o. Reacts with second equivalent of alcohol (sn1) reaction to yield acetal. Acid protonates oh excellent lg carbocation alcohol acts as nucleophile. Reversible acetal reaction with protonation of or (good.