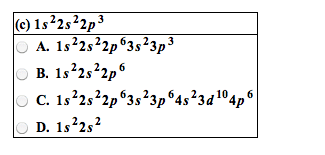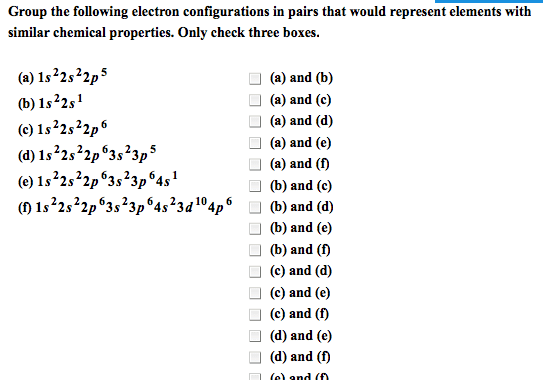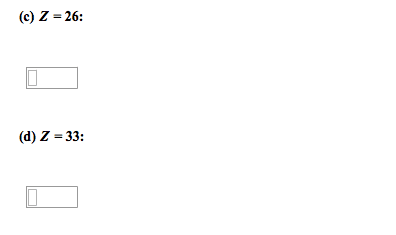CHEM 1E03 Chapter Notes - Chapter 4: Noble Gas, Calcium Hydroxide, Electron Configuration

63
CHEM 1E03 Full Course Notes
Verified Note
63 documents
Document Summary
Concepts: review ib notes, metals and non-metals and their ions. Ionization energy: electron affinity, magnetic properties, periodic properties of the elements, electronegativity. Noble gases, group 18 (8a: until 1962, noble gases were not thought to undergo any chemical reactions and were considered inert. In 1962, neil bartlett discovered that some noble gases react. Noble gases since then have been synthesized for the heavier elements of the family, namely krypton and xenon. Trends in non-metals: non-metals that lack 1,2, or 3 electrons fill their shells by gaining electrons to try to become like noble gases and gain stability. The high affinity that halogens have for electrons means that when you react them with elements like alkali metals, vigorous reactions ensue. Non-metal oxides: when non-metal reacts with oxygen, the resulting oxides have covalent bonding. If we think of sulfur, a can burn it in the presence of a catalyst, and drive it all the way to the oxide, so3.