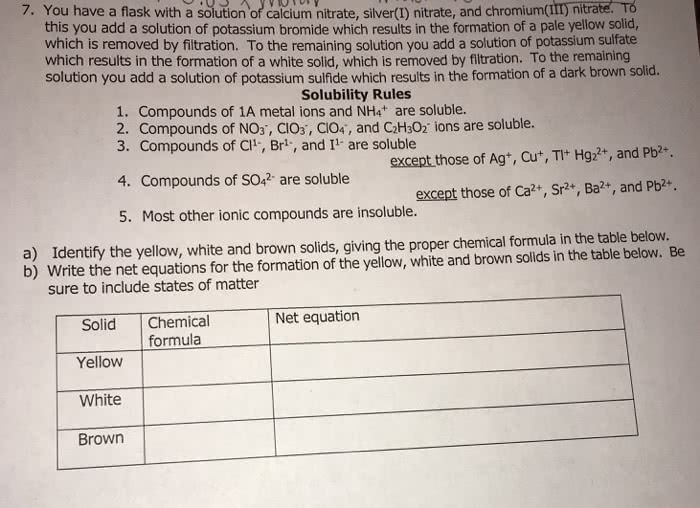
CHEM 24112 Study Guide - Quiz Guide: Nickel(Ii) Chloride, Barium Nitrate, Potassium Sulfate

Practice Test #3
Multiple Choice
Identify the choice that best completes the statement or answers the question.
____ 1. The factors that most commonly cause chemical reactions to occur are all the following EXCEPT
a. formation of a solid c. transfer of electrons
b. formation of a gas d. a decrease in temperature
____ 2. When a precipitation reaction occurs, the ions that do NOT form the precipitate
a. evaporate c. form a second insoluble compound in
solution
b. are cations only d. are left dissolved in the solution
____ 3. An aqueous solution of potassium sulfate is allowed to reat with an aqueous solution of barium nitrate. What
is the coefficient of the solid in the balanced equation?
a. 1 b. 2 c. 3 d. 4
____ 4. An aqueous solution of sodium sulfate is allowed to react wtih an aqueous solution of calcium nitrate. The
complete ionic equation contains which of the following species?
a. 2Na+b. 2SO42- c. 3Ca2+ d. NO3-
____ 5. An aqueous solution of ammonium carbonate is allowed to react with an aqueous solution of nickel(II)
chloride. Identify the solid in the balanced equation.
a. NiCO3b. NH4Cl c. NiCl2d. (NH4)2CO3
____ 6. When the following equation is balanced, what is the coefficient for H2O?
Ca(OH)2 + H3PO4 --> Ca3(PO4)2 + H2O
a. 2 b. 3 c. 4 d. 6
____ 7. In what type of reaction is water always a product?
a. precipitation c. oxidation
b. acid-base d. decomposition
____ 8. The equation 2Ag2O(s) --> 4Ag(s) + O2(g) is a(n) _____ reaction.
a. oxidation reduction only c. synthesis
b. decomposition only d. oxidation reduction and decomposition
____ 9. The equation CH4(g) + 2O2(g) --> CO2(g) + 2H2O(g) is a(n) ___ reaction.
a. synthesis c. combustion
b. precipitation d. decomposition
____ 10. Which reaction pattern does the following reaction fit? 2HgO(s) --> 2Hg(l) + O2(g)
a. synthesis c. single replacement
b. decomposition d. exchange
____ 11. All of the following are clues that a chemical reaction has taken place EXCEPT
a. a color change occurs c. the reactant is smaller
b. a solid forms d. bubbles form
____ 12. When the following equation is balanced, what is the number in front of the substance in bold type? Na2S2O3
+ I2 --> NaI + Na2S4O6
a. 1 b. 2 c. 3 d. 4
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Identify the choice that best completes the statement or answers the question. ___: the factors that most commonly cause chemical reactions to occur are all the following except a. b. formation of a solid formation of a gas transfer of electrons c, a decrease in temperature. ___: when a precipitation reaction occurs, the ions that do not form the precipitate a. evaporate, are cations only c. form a second insoluble compound in solution, are left dissolved in the solution. ___: an aqueous solution of potassium sulfate is allowed to reat with an aqueous solution of barium nitrate. What is the coefficient of the solid in the balanced equation: 1, 2, 3, 4. ___: an aqueous solution of sodium sulfate is allowed to react wtih an aqueous solution of calcium nitrate. The complete ionic equation contains which of the following species: 2na, 3ca2, 2so4.