CHEM 1202 : SI Exam 1 Review
Document Summary
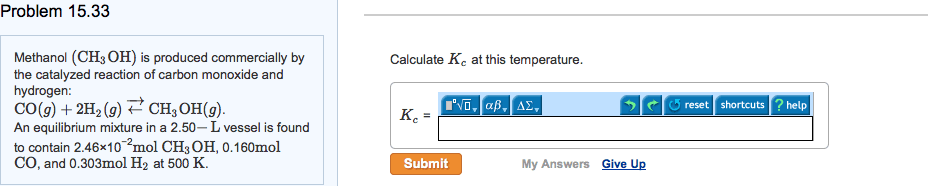
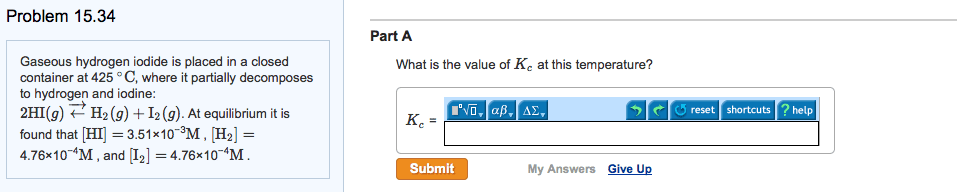
Write the equilibrium constant for the heterogeneous reaction 2nahco3(s) na2co3(s) + co2(g) + H2o(g): gaseous hydrogen iodide is placed in a closed 1. 0-l container at 425 c, where it partially decomposes to hydrogen and iodine: At equilibrium it is found that phi = 3. 53 10 3 atm; ph2 = 4. 79 10 4 atm; and pi2 = 4. 79 . What is the value of kp at this temperature: at 100 c the equilibrium constant for the reaction cocl2(g) co(g) + cl2(g) has a value of kp = 2. 19 . At some constant temperature, the kp vaule for this reaction is 0. 16. Find the equilibrium partial pressures for all three gases. Suppose that initially, the pressure of brcl is 7. 39x10-3, and there is no br2 or cl2 present. Calculate the equilibrium partial pressure of br2: gaseous so3 is placed in a closed container at 602 c, where it partially decomposes to so2 and o2: