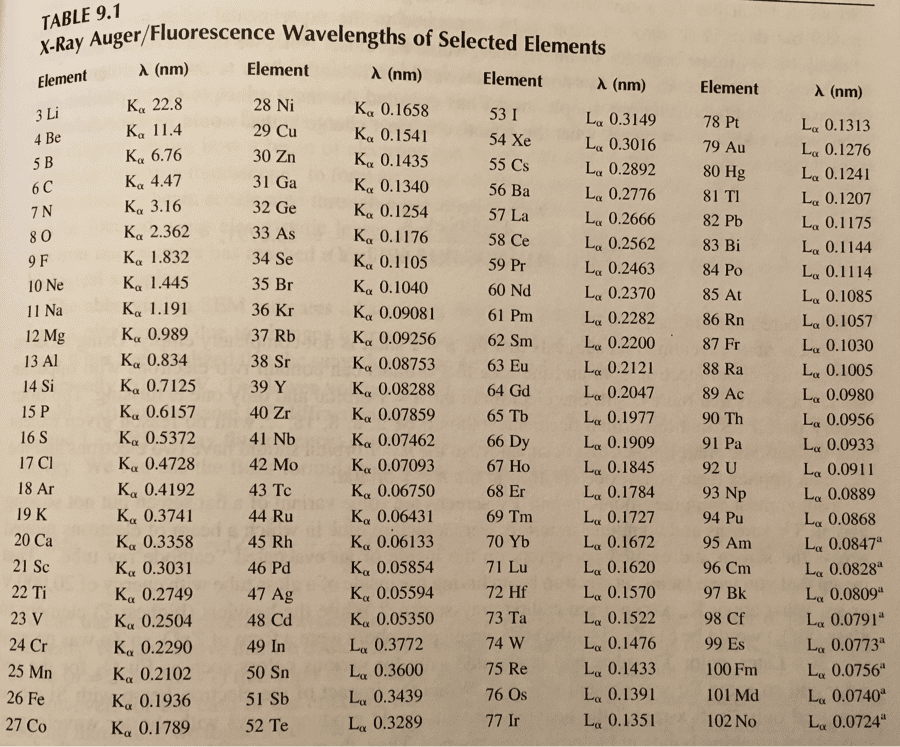
CHM 1321 Study Guide - Final Guide: Fractional Distillation, Distillation, Boiling Point
Get access

Related Documents
Related Questions
| Temp | %A in Vapor | %A in Liquid |
| 70 | 100 | 100 |
| 75 | 97 | 79 |
| 80 | 93 | 65 |
| 85 | 85 | 55 |
| 90 | 77 | 45 |
| 95 | 69 | 37 |
| 100 | 60 | 28 |
| 105 | 50 | 20 |
| 110 | 40 | 12 |
| 115 | 25 | 6 |
| 120 | 0 | 0 |
The table above shows the % compositions of the vapor and liquid phases at equilibrium for solutions of A and B at the indicated temperatures:
1). A mixture of 30g of A and 70g of B is distilled using a dimple distillation apparatus. At what temperature would the first drop distill?
2). A misture of 50g of A and 50g of B is distilled usinga very efficient fractionating column. Construct a distillation curve (temperature vs. mL of distillate) for this system knowing that the density of A and B is 1.05g/mL and 0.96g/mL, respectively.
Give It Some Thought 6.16
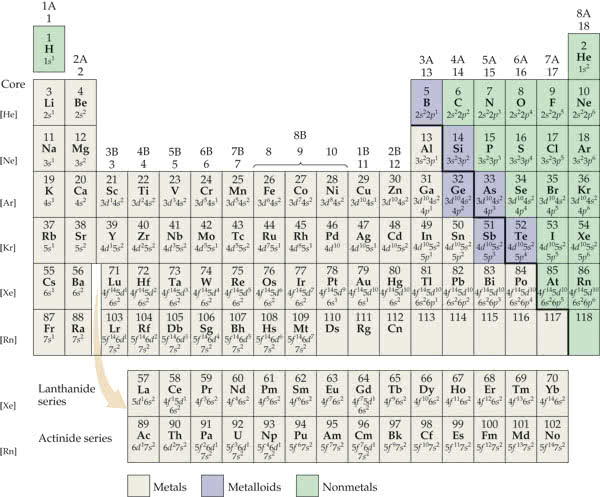
Valence electron configurations of the elements.
Part A
The elements Ni, Pd, and Pt are all in the same group. By examining the electron configurations for these elements in the following figure, what can you conclude about the relative energies of the nd and (n+1)s orbitals for this group?
The elements Ni, Pd, and Pt are all in the same group. By examining the electron configurations for these elements in the following figure, what can you conclude about the relative energies of the and orbitals for this group?
| nd > (n+1)s |
| nd < (n+1)s |
| nd = (n+1)s |
| It is impossible to conclude. |