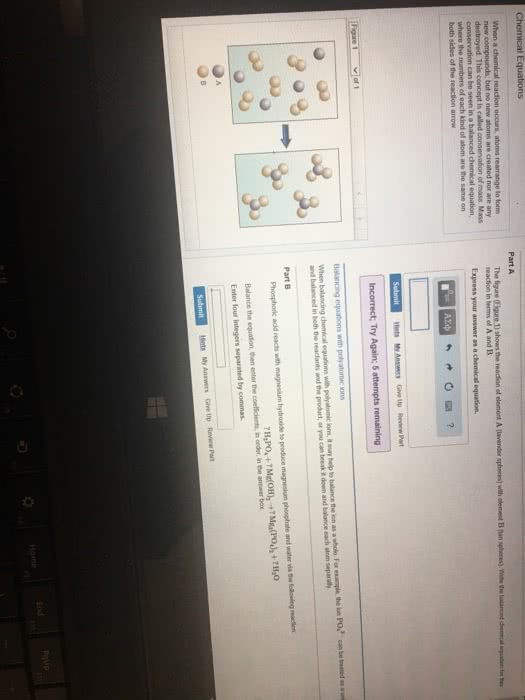
2
answers
0
watching
43
views
18 Dec 2019
Which of the following statements is not true of balancing a chemical equation?
a. Subscripts in the reactants must be conserved in the products.
b. Coefficients are used to balance the atoms on both sides.
c. Phases are often shown for each compound but are not critical to balancing an equation.
d. The law of conservation of matter must be followed.
e. All of the above statements (a-d) are true.
Which of the following statements is not true of balancing a chemical equation?
| a. | Subscripts in the reactants must be conserved in the products. | |
| b. | Coefficients are used to balance the atoms on both sides. | |
| c. | Phases are often shown for each compound but are not critical to balancing an equation. | |
| d. | The law of conservation of matter must be followed. | |
| e. | All of the above statements (a-d) are true. |
23 Nov 2021
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Reid WolffLv2
31 Dec 2019
Get unlimited access
Already have an account? Log in