1
answer
0
watching
1,860
views
6 Oct 2020
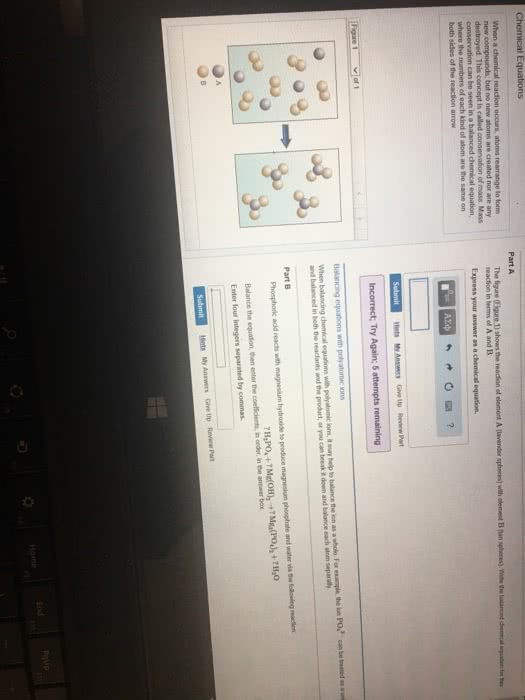
Which of the following statements is true about chemical reactions?
a. When balancing a chemical equation, you can never change the coefficient in front of any chemical formula.
b. The coefficients in a balanced chemical equation refer to the number of grams of reactants and products.
c. In a chemical equation, the reactants are on the right and the products are on the left.
d. When balancing a chemical equation, you can never change the subscripts of any chemical formula.
e. In chemical reactions, matter is neither created nor destroyed so a chemical equation must have the same number of atoms on both sides of the equation.
Which of the following statements is true about chemical reactions?
a. When balancing a chemical equation, you can never change the coefficient in front of any chemical formula.
b. The coefficients in a balanced chemical equation refer to the number of grams of reactants and products.
c. In a chemical equation, the reactants are on the right and the products are on the left.
d. When balancing a chemical equation, you can never change the subscripts of any chemical formula.
e. In chemical reactions, matter is neither created nor destroyed so a chemical equation must have the same number of atoms on both sides of the equation.
Matt OcampoLv10
8 Dec 2020