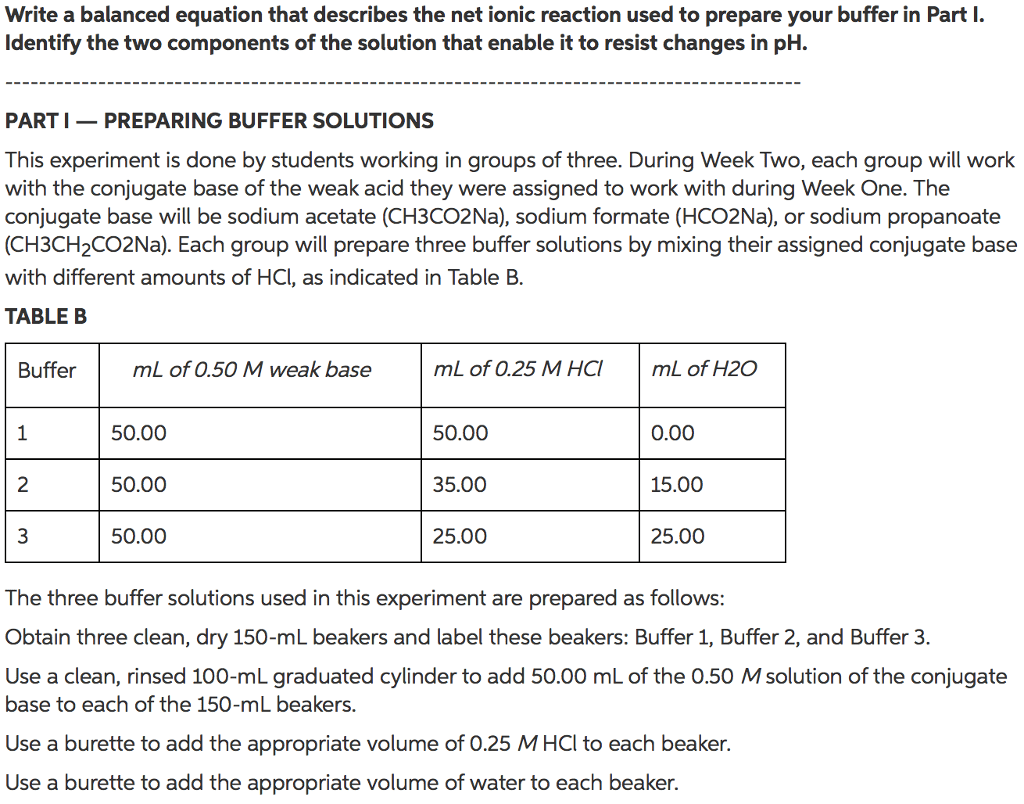
1. Label four clean 100 and/or 150 mL beakers: A, B, C, and D.
2. Add 40 mL of distilled water to beakers A and C. Be sure that the graduated cylinder is well rinsed before using it.
3. Use a graduated cylinder to add 40 mL of the prepared sodium acetate/acetic acid buffer solution to both beakers B and D.
4. Record the initial pH of each the solutions A-D. Make sure to rinse the pH electrode with distilled water between readings.
5. Pipette 1.0 mL of 6 M HCl, hydrochloric acid solution into beakers A and B. Thoroughly mix the solutions.
6. Pipette 1.0 mL of 6 M NaOH, sodium hydroxide solution into beakers C and D. Thoroughly mix the solutions
What is the volume of 6HCl used for addition into beaker A,B,C,D and how do you calculate the ph of each beaker after the addition of 6M HCL
1. Label four clean 100 and/or 150 mL beakers: A, B, C, and D.
2. Add 40 mL of distilled water to beakers A and C. Be sure that the graduated cylinder is well rinsed before using it.
3. Use a graduated cylinder to add 40 mL of the prepared sodium acetate/acetic acid buffer solution to both beakers B and D.
4. Record the initial pH of each the solutions A-D. Make sure to rinse the pH electrode with distilled water between readings.
5. Pipette 1.0 mL of 6 M HCl, hydrochloric acid solution into beakers A and B. Thoroughly mix the solutions.
6. Pipette 1.0 mL of 6 M NaOH, sodium hydroxide solution into beakers C and D. Thoroughly mix the solutions
What is the volume of 6HCl used for addition into beaker A,B,C,D and how do you calculate the ph of each beaker after the addition of 6M HCL