timothystark
Timothy Anthony
2 Followers
0 Following
0 Helped
timothystarkLv9
5 Jan 2023
Answer:One of the main tools The Fed uses to fix inflation is raising interest...
timothystarkLv9
1 Dec 2022
Answer:7. We have to determine the weight that represents 58th percentile Give...
timothystarkLv9
1 Dec 2022
Answer:Given that , n = 47 x = 30 The null and alternative hypothesis is H0 : ...
timothystarkLv9
4 Nov 2022
Answer: Types of Chemical Reactions Balance each of the following reactions an...
timothystarkLv9
3 Nov 2022
Answer: 4) While running PCR wearing gloves is necessary . The reason being is...
timothystarkLv9
3 Nov 2022
Answer: The reaction between phenolphthalein and NaOH is first order in respec...
timothystarkLv9
2 Nov 2022
Answer: 4) While running PCR wearing gloves is necessary . The reason being is...
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer:When water vapor condenses it adds heat to the wall of the cup. That sa...
timothystarkLv9
2 Nov 2022
Answer: Pressure of vapor = 766.32 0.00131 = 1.0038 atm For the volume of the ...
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
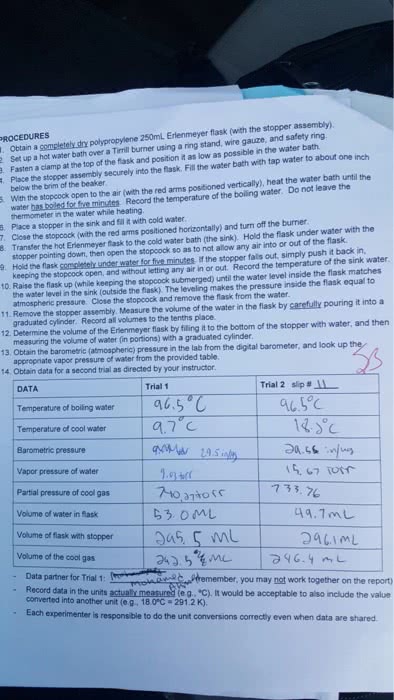
Answer: Sample calculation for trial 1 : Atmospheric pressure = 760 mmHg Press...
timothystarkLv9
2 Nov 2022
Answer: Trial 1 Trial 2 1) Mass of flask and stopper (g) 148.866 148.866 2) Ma...
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer: 1. For the gas, a. pressure of gas = 728/760 = 0.96 atm b. Temperature...
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer:flask = 55.844g5ml liquid at 99.7ºCmass flask after heating = 56.101gvo...
timothystarkLv9
2 Nov 2022
Answer: flask = 54.868g 5ml liquid at 100ºC mass flask after heating = 55.496g...
timothystarkLv9
2 Nov 2022
Answer: Solution:- Given data is... mass of empty flask and rubber stopper = 8...
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer: The sublimation process involved loss of CO2 as gas from the flask. Th...
timothystarkLv9
2 Nov 2022
Answer: The sublimation process involved loss of CO2 as gas from the flask. Th...
timothystarkLv9
2 Nov 2022
Answer: 1. so that all the other gases could escape away from the flask and on...
timothystarkLv9
2 Nov 2022
Answer: 1) Excess dry ice was used to ensure that significant loss of CO2 does...
timothystarkLv9
2 Nov 2022
Answer: Part 1. moles of sodium acetate trihydrate = g/molar mass = 3.3/136.08...
timothystarkLv9
2 Nov 2022
Answer: moles of sodium acetate trihydrate = 3.3 / 136.08 = 0.024 molarity = m...
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer: moles of sodium acetate trihydrate = 3.3 / 136.08 = 0.024 molarity = m...
timothystarkLv9
2 Nov 2022
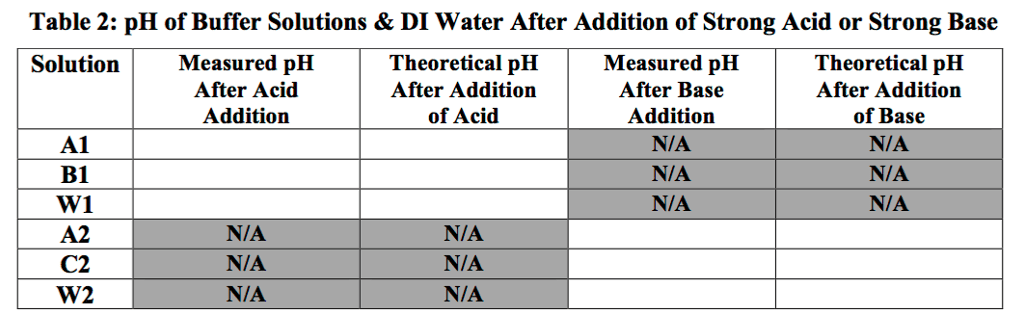
Answer: pH of solution adding 0.10 M HCl to 100 mL water HCl added pH 0 mL 7.0...
timothystarkLv9
2 Nov 2022
Answer: American Indians and Alaska Natives have consistently experienced disp...
timothystarkLv9
2 Nov 2022
Answer: I need the pKa value for this buffer, do you have it?. I found this, b...
timothystarkLv9
2 Nov 2022
Answer:
timothystarkLv9
2 Nov 2022
Answer:Experiment5: Acid-Base Equilibria, pH, and BuffersPurpose:The purpose o...
timothystarkLv9
2 Nov 2022
Answer:Experiment5: Acid-Base Equilibria, pH, and BuffersPurpose:The purpose o...
timothystarkLv9
2 Nov 2022
Answer:Using a funnel, transfer the sodium carbonate into a clean, dry 250mL ....
timothystarkLv9
2 Nov 2022
Answer: Test tube colour observed volume in the tube at the end 1. Red 15mL 2....
timothystarkLv9
2 Nov 2022
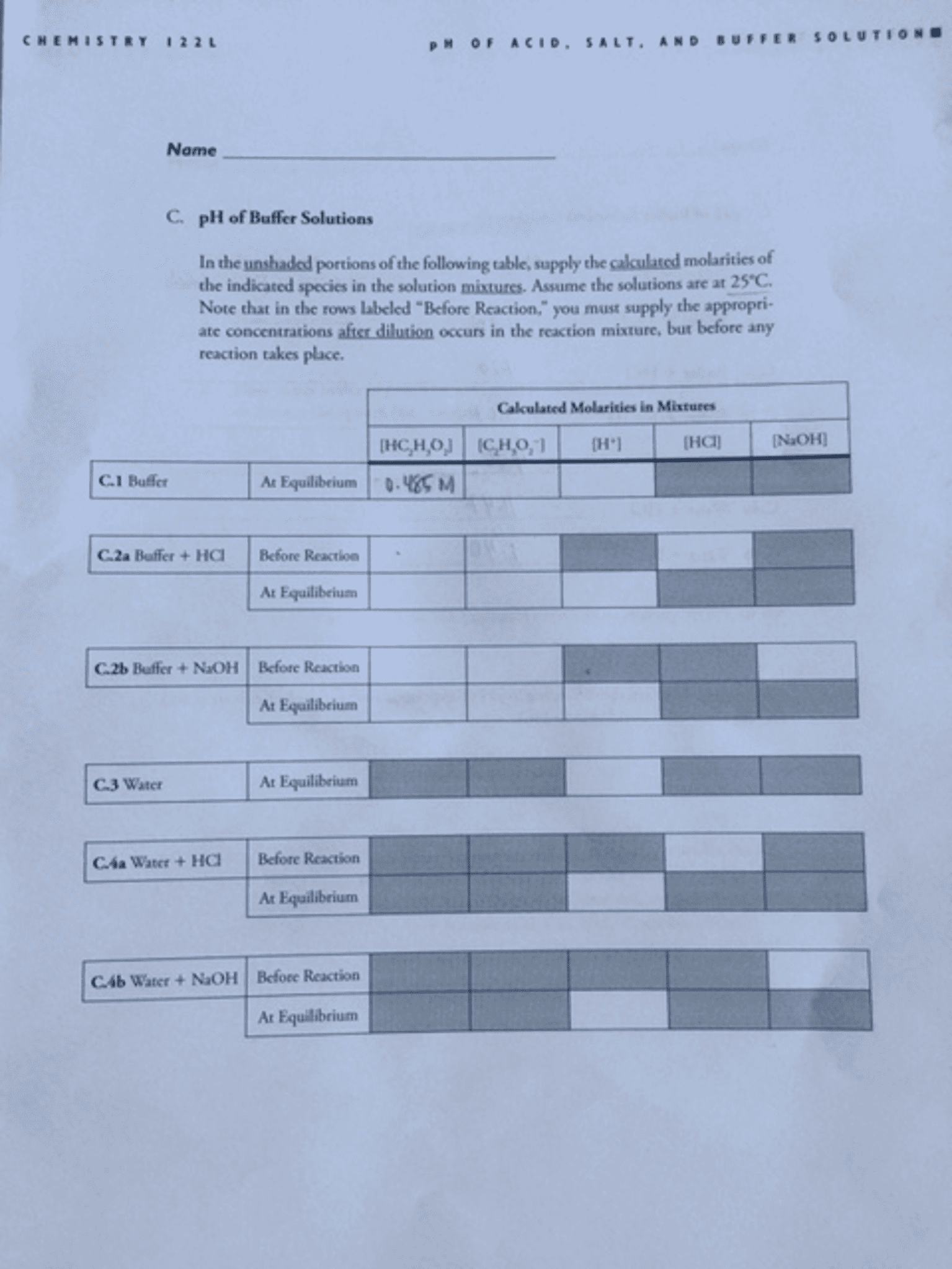
Answer:Using Hendeerson-Hasselbalch equation:PH = Pka + log [CH3COO-]/[CH3COOH...
timothystarkLv9
2 Nov 2022
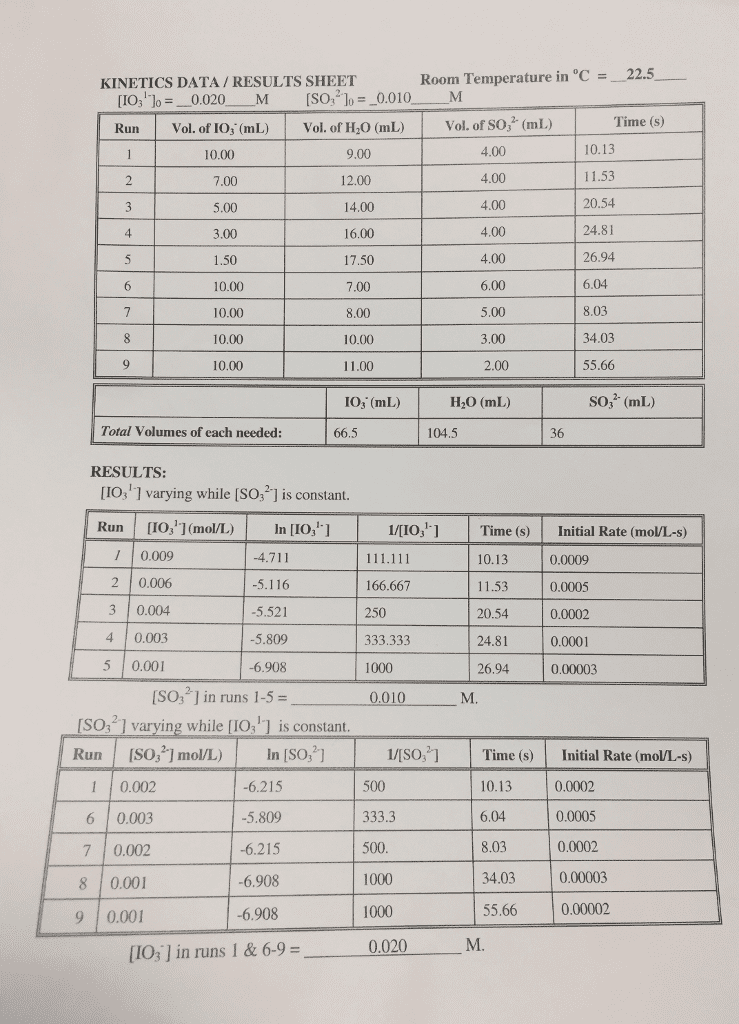
Answer: Therefore, k = Rate / [IO3-]1 [SO32-]2 k = 0.0002 / (0.004) (0.010)2 k...