1
answer
0
watching
280
views
blackcrab245Lv1
1 Apr 2020
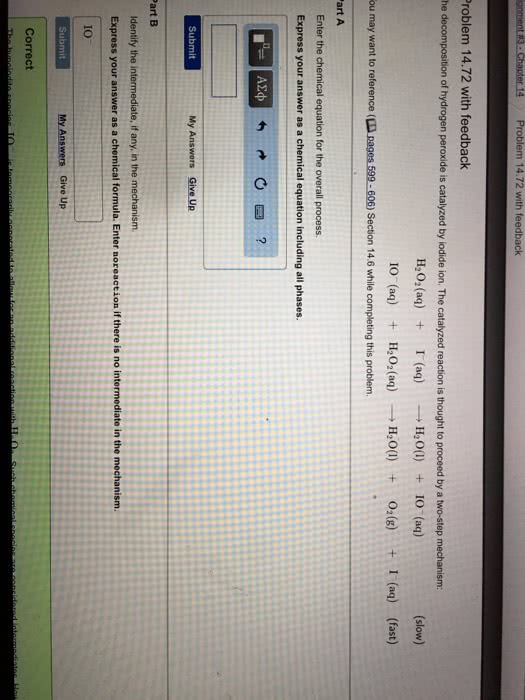
The decomposition of hydrogen peroxide is catalyzed by iodide ion. The catalyzed reaction is thought to proceed by a two-step mechanism:
H2O2(aq) + I– (aq)  H2O(l) + IO– (aq) (slow)
H2O(l) + IO– (aq) (slow)
IO– (aq) + H2O2(aq)  H2O(l) + O2(g) + I– (aq) (fast)
H2O(l) + O2(g) + I– (aq) (fast)
(a) Write the chemical equation for the overall process. (b) Identify the intermediate, if any, in the mechanism. (c) Assuming that the first step of the mechanism is rate determining, predict the rate law for the overall process.
The decomposition of hydrogen peroxide is catalyzed by iodide ion. The catalyzed reaction is thought to proceed by a two-step mechanism:
H2O2(aq) + I– (aq) H2O(l) + IO– (aq) (slow)
IO– (aq) + H2O2(aq) H2O(l) + O2(g) + I– (aq) (fast)
(a) Write the chemical equation for the overall process. (b) Identify the intermediate, if any, in the mechanism. (c) Assuming that the first step of the mechanism is rate determining, predict the rate law for the overall process.
Bunny GreenfelderLv2
27 May 2020