2
answers
0
watching
257
views
23 Nov 2019
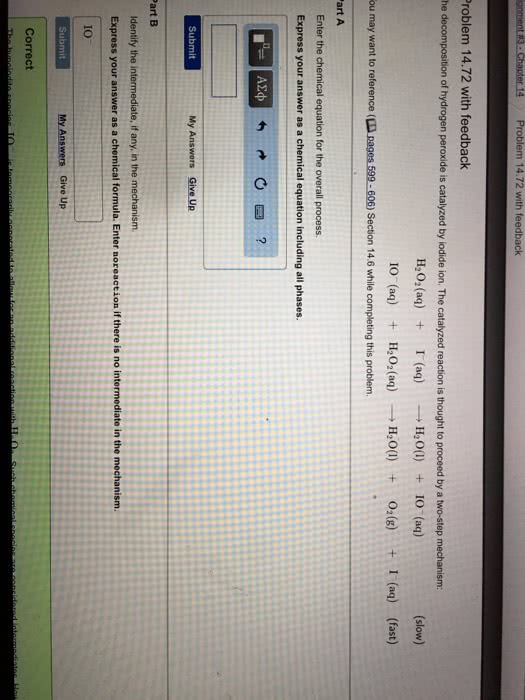
The decomposition of hydrogen peroxide is catalyzed by iodide ion.The catalyzed reaction is thought to proceed by a two mechanismreaction
H2O2(aq) + I-(aq)--->H2O(l)+lO-(aq) (SLOW)
H2O(l)+lO-(aq)--->H2O(aq) + O2 + I-(aq) (fast)
A. write the rate law for the overall reaction.
B. Write the chemical equation for the overall process and identifythe intermediate if any.
The decomposition of hydrogen peroxide is catalyzed by iodide ion.The catalyzed reaction is thought to proceed by a two mechanismreaction
H2O2(aq) + I-(aq)--->H2O(l)+lO-(aq) (SLOW)
H2O(l)+lO-(aq)--->H2O(aq) + O2 + I-(aq) (fast)
A. write the rate law for the overall reaction.
B. Write the chemical equation for the overall process and identifythe intermediate if any.
Beverley SmithLv2
18 Aug 2019
Already have an account? Log in