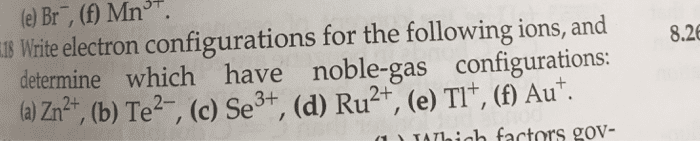1
answer
1
watching
255
views
jadebear386Lv1
19 Mar 2020
Write the electron configurations for the following ions, and determine which have noble-gas configurations: (a) Ru3 +, (b) As3-, (c) Y3 +, (d) Pd2+, (e) Pb2+, (f) Au3+.
Write the electron configurations for the following ions, and determine which have noble-gas configurations: (a) Ru3 +, (b) As3-, (c) Y3 +, (d) Pd2+, (e) Pb2+, (f) Au3+.
Collen VonLv2
24 May 2020