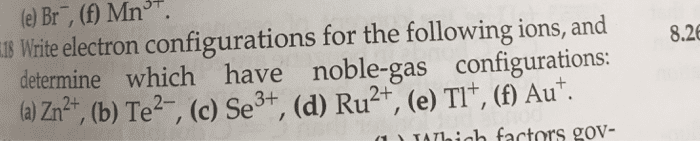1
answer
0
watching
114
views
13 Dec 2019
Write electron configurations for the following ions, and determine which have noble-gas configurations. (Type your answer using the format [Ar] 4s2 3d10 4p2 for [Ar]4s23d104p2. If the configuration is a noble gas, enter the noble gas in brackets, for example [Ne] for F -.)
(a) Tl+
(b) Ni2+
(c) Se3+
(d) In2+
(e) Ag+
(f) Sn2+
Write electron configurations for the following ions, and determine which have noble-gas configurations. (Type your answer using the format [Ar] 4s2 3d10 4p2 for [Ar]4s23d104p2. If the configuration is a noble gas, enter the noble gas in brackets, for example [Ne] for F -.)
(a) Tl+
(b) Ni2+
(c) Se3+
(d) In2+
(e) Ag+
(f) Sn2+
Irving HeathcoteLv2
17 Dec 2019