1
answer
0
watching
279
views
13 Dec 2019
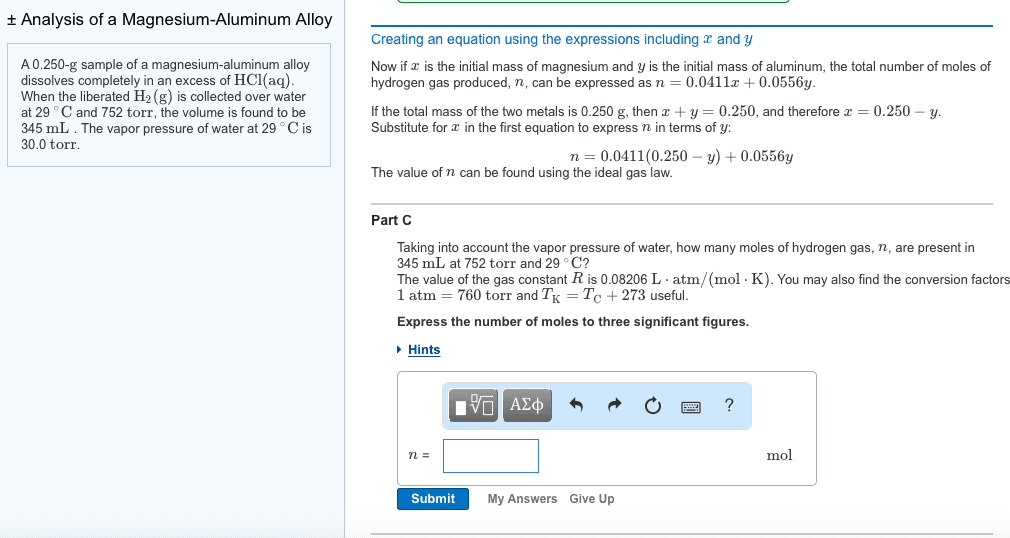
A 0.250-g sample of a magnesium-aluminum alloy dissolvescompletely in an excess of HCl(aq). When the liberated H2(g) iscollected over water at 29 ?C and 752 torr, the volume is found tobe 313mL . The vapor pressure of water at 29 ?C is 30.0 torr.
How many moles of H2 can be produced from y grams of Al inmagnesium-aluminum alloy? The molar mass of Al is 26.98 g/mol.
A 0.250-g sample of a magnesium-aluminum alloy dissolvescompletely in an excess of HCl(aq). When the liberated H2(g) iscollected over water at 29 ?C and 752 torr, the volume is found tobe 313mL . The vapor pressure of water at 29 ?C is 30.0 torr.
How many moles of H2 can be produced from y grams of Al inmagnesium-aluminum alloy? The molar mass of Al is 26.98 g/mol.
Bunny GreenfelderLv2
17 Dec 2019