1
answer
0
watching
316
views
28 Sep 2019
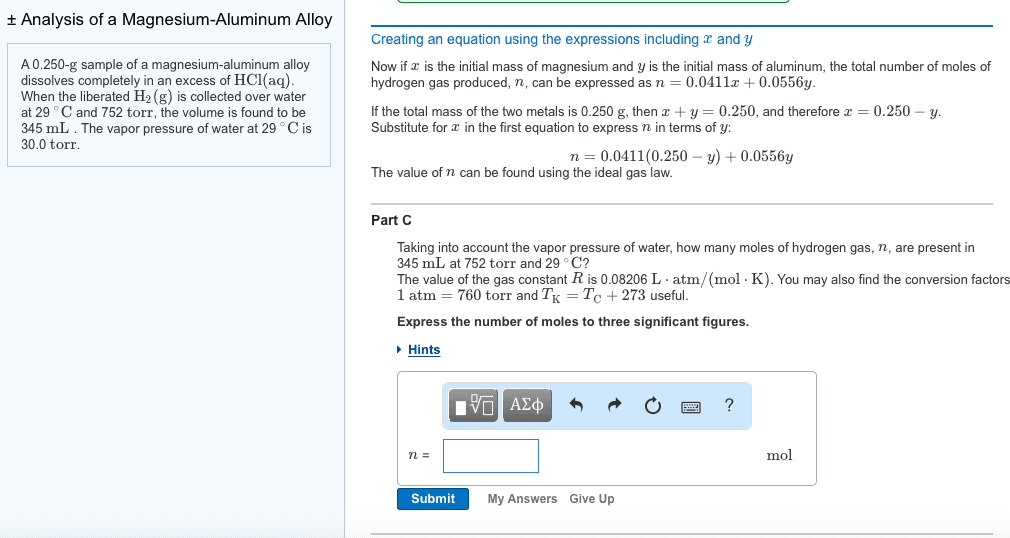
A 0.250- sample of a magnesium-aluminum alloy dissolves completely in an excess of . When the liberated is collected over water at 29 and 752 , the volume is found to be 305 . The vapor pressure of water at 29 is 30.0 How many moles of can be produced from grams of in magnesium-aluminum alloy? The molar mass of is 24.31 . Express your answer in terms of to four decimal places (i.e., 0.5000x).
A 0.250- sample of a magnesium-aluminum alloy dissolves completely in an excess of . When the liberated is collected over water at 29 and 752 , the volume is found to be 305 . The vapor pressure of water at 29 is 30.0 How many moles of can be produced from grams of in magnesium-aluminum alloy? The molar mass of is 24.31 . Express your answer in terms of to four decimal places (i.e., 0.5000x).
Jarrod RobelLv2
28 Sep 2019