1
answer
0
watching
385
views
13 Dec 2019
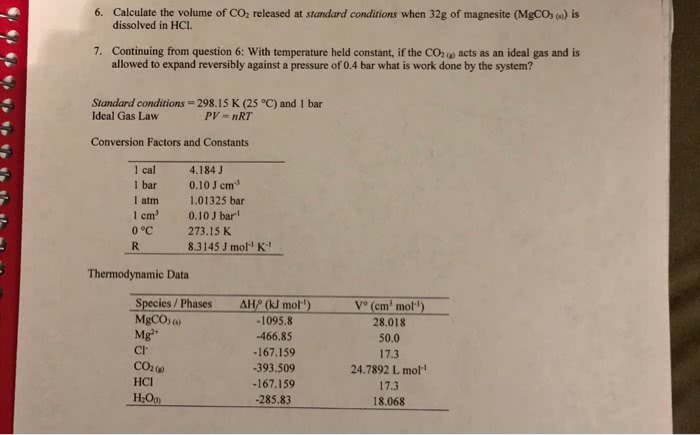
A 65.56 g quantity of dry ice (solid carbon dioxide) is allowed to sublime (evaporate) in a position like apparatus. Calculate the expansion work done against external pressure of 0.956 atm and at a constant temperature of 22C. Assume that the initial volume of dry ice id negligible and the CO2 behaves as an ideal gas (1Lâ¢atm=101.3J)
A 65.56 g quantity of dry ice (solid carbon dioxide) is allowed to sublime (evaporate) in a position like apparatus. Calculate the expansion work done against external pressure of 0.956 atm and at a constant temperature of 22C. Assume that the initial volume of dry ice id negligible and the CO2 behaves as an ideal gas (1Lâ¢atm=101.3J)
1
answer
0
watching
385
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Irving HeathcoteLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232