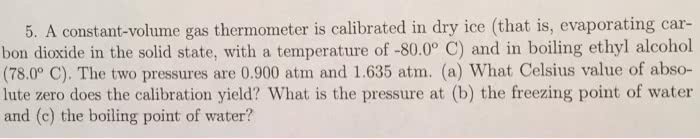1
answer
0
watching
99
views
13 Dec 2019
A 17.5-g quantity of dry ice (solid carbon dioxide) is allowed to sublime (evaporate) in an apparatus like the one shown in the figure below. Calculate the expansion work done against a constant external pressure of 0.975 atm and at a constant temperature of 26°C. Assume that the initial volume of dry ice is negligible and that CO2 behaves like an ideal gas.
A 17.5-g quantity of dry ice (solid carbon dioxide) is allowed to sublime (evaporate) in an apparatus like the one shown in the figure below. Calculate the expansion work done against a constant external pressure of 0.975 atm and at a constant temperature of 26°C. Assume that the initial volume of dry ice is negligible and that CO2 behaves like an ideal gas.
Tod ThielLv2
17 Dec 2019