9
answers
0
watching
1,126
views
26 Jun 2018
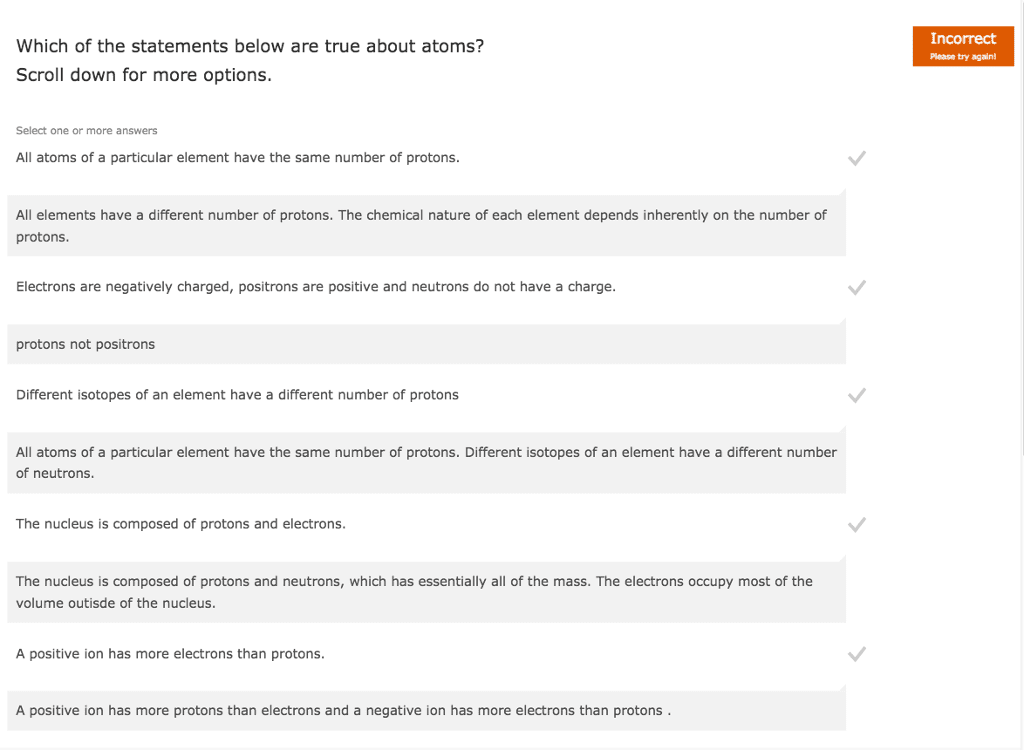
Which of the following statements is TRUE? (a) The atomic number is the same as the mass number. (b) The atomic number is the same as the number of protons. (c) The identity of a chemical element is determined by the number of valence electrons. (d) The radius of all atoms can be measured precisely. (e) Different isotopes of an element have different atomic numbers.
Which of the following statements is TRUE? (a) The atomic number is the same as the mass number. (b) The atomic number is the same as the number of protons. (c) The identity of a chemical element is determined by the number of valence electrons. (d) The radius of all atoms can be measured precisely. (e) Different isotopes of an element have different atomic numbers.
Read by 1 person
lalithashwin156Lv10
30 May 2023
Read by 1 person
akunuru639Lv10
28 May 2023
Already have an account? Log in
Read by 1 person
Read by 1 person
Read by 1 person
Read by 1 person
Read by 1 person
Read by 1 person
Deanna HettingerLv2
28 Jun 2018
Already have an account? Log in